Global Machine Learning in Pharmaceuticals Market - Key Trends & Drivers Summarized
Why Are Drug Development Pipelines Becoming Algorithm Guided?
Pharmaceutical research is transitioning from sequential experimentation toward data guided discovery as molecular databases and biological datasets expand rapidly. Traditional compound screening relied on laboratory iteration across thousands of molecules, but machine learning models now predict binding affinity, toxicity probability, and pharmacokinetic behavior before synthesis occurs. Research teams integrate genomic data, protein structure repositories, and historical clinical outcomes to identify candidate molecules with higher probability of therapeutic success. Predictive modeling narrows chemical libraries into focused candidates that match target receptors and biological pathways, significantly reshaping early stage discovery workflows. Formulation development has also adopted predictive modeling to estimate solubility and stability characteristics under different storage conditions. Instead of repeated laboratory trials, simulation driven evaluation determines excipient combinations and dosage delivery mechanisms. As a result, drug discovery cycles increasingly combine computational hypothesis generation with targeted laboratory validation rather than broad exploratory testing.How Is Clinical Research Being Redesigned Around Patient Data Patterns?
Clinical trials are being reorganized using patient stratification models that analyze medical history, genetic markers, and treatment response variability. Rather than enrolling large undifferentiated populations, machine learning algorithms identify patient subgroups most likely to respond to a therapy, improving outcome clarity and reducing trial size requirements. Recruitment platforms analyze electronic health records and diagnostic imaging metadata to match eligible participants automatically, accelerating enrollment timelines. During trials, predictive safety monitoring evaluates vital signs, lab results, and reported symptoms to anticipate adverse events before clinical escalation occurs. Adaptive trial designs increasingly adjust dosage levels and cohort composition using model generated probability estimates of treatment efficacy. Real world evidence studies also leverage prescription patterns and hospitalization data to evaluate therapy performance after approval. This approach transforms clinical research from static study protocols into continuously monitored experiments guided by incoming patient data streams.Are Manufacturing And Quality Control Moving Toward Predictive Compliance?
Pharmaceutical production environments are adopting machine learning to monitor process variability and maintain batch consistency. Manufacturing lines generate sensor data related to temperature stability, mixing duration, and pressure conditions during synthesis and packaging. Models detect subtle deviations that historically would be identified only during end stage quality testing. Predictive release systems evaluate in process measurements to determine whether a batch meets quality thresholds before final inspection. Packaging verification also uses image recognition models to confirm labeling accuracy and detect microscopic defects in vials and tablets. Supply chain integrity monitoring applies predictive analysis to storage temperature logs and transportation duration to determine potential product degradation risk. Regulatory compliance therefore shifts from retrospective testing toward continuous assurance throughout the manufacturing lifecycle.What Forces Are Expanding Adoption Across The Pharmaceutical Value Chain?
The growth in the machine learning in pharmaceuticals market is driven by several factors including increasing complexity of biologic drug discovery requiring molecular prediction models, rising clinical trial costs encouraging targeted patient recruitment and adaptive study designs, expansion of real world evidence datasets from electronic health records enabling post market analysis, regulatory expectations for continuous manufacturing monitoring, demand for faster vaccine and therapy development cycles, growth of personalized medicine requiring genotype based treatment selection, need for predictive pharmacovigilance using large adverse event databases, increasing outsourcing to contract research organizations that depend on automated data screening, and global cold chain distribution challenges that require predictive stability assessment during transportation.Report Scope
The report analyzes the ML in Pharmaceuticals market, presented in terms of market value (US$). The analysis covers the key segments and geographic regions outlined below:- Segments: Technology (Supervised Learning Technology, Deep Learning Technology, Unsupervised Learning Technology, Reinforcement Learning Technology, Other Technologies); Deployment (Cloud Deployment, Hybrid Deployment, On-Premise Deployment); Application (Drug Discovery & Design Application, Clinical Trial Optimization Application, Disease Diagnosis & Biomarker Identification Application, Manufacturing & Quality Control Application, Other Applications)
- Geographic Regions/Countries: World; USA; Canada; Japan; China; Europe; France; Germany; Italy; UK; Rest of Europe; Asia-Pacific; Rest of World.
Key Insights:
- Market Growth: Understand the significant growth trajectory of the Supervised Learning Technology segment, which is expected to reach US$7.4 Billion by 2032 with a CAGR of a 31.3%. The Deep Learning Technology segment is also set to grow at 36.4% CAGR over the analysis period.
- Regional Analysis: Gain insights into the U.S. market, valued at $1.1 Billion in 2025, and China, forecasted to grow at an impressive 34.7% CAGR to reach $5.1 Billion by 2032. Discover growth trends in other key regions, including Japan, Canada, Germany, and the Asia-Pacific.
Why You Should Buy This Report:
- Detailed Market Analysis: Access a thorough analysis of the Global ML in Pharmaceuticals Market, covering all major geographic regions and market segments.
- Competitive Insights: Get an overview of the competitive landscape, including the market presence of major players across different geographies.
- Future Trends and Drivers: Understand the key trends and drivers shaping the future of the Global ML in Pharmaceuticals Market.
- Actionable Insights: Benefit from actionable insights that can help you identify new revenue opportunities and make strategic business decisions.
Key Questions Answered:
- How is the Global ML in Pharmaceuticals Market expected to evolve by 2032?
- What are the main drivers and restraints affecting the market?
- Which market segments will grow the most over the forecast period?
- How will market shares for different regions and segments change by 2032?
- Who are the leading players in the market, and what are their prospects?
Report Features:
- Comprehensive Market Data: Independent analysis of annual sales and market forecasts in US$ Million from 2025 to 2032.
- In-Depth Regional Analysis: Detailed insights into key markets, including the U.S., China, Japan, Canada, Europe, Asia-Pacific, Latin America, Middle East, and Africa.
- Company Profiles: Coverage of players such as AbbVie, Inc., Amgen, Inc., AstraZeneca Plc, Bayer AG, Bristol-Myers Squibb Company and more.
- Complimentary Updates: Receive free report updates for one year to keep you informed of the latest market developments.
Some of the companies featured in this ML in Pharmaceuticals market report include:
- AbbVie, Inc.
- Amgen, Inc.
- AstraZeneca Plc
- Bayer AG
- Bristol-Myers Squibb Company
- Eli Lilly and Company
- F. Hoffmann-La Roche Ltd.
- GSK Plc
- ICON Plc
- Insilico Medicine
Domain Expert Insights
This market report incorporates insights from domain experts across enterprise, industry, academia, and government sectors. These insights are consolidated from multilingual multimedia sources, including text, voice, and image-based content, to provide comprehensive market intelligence and strategic perspectives. As part of this research study, the publisher tracks and analyzes insights from 43 domain experts. Clients may request access to the network of experts monitored for this report, along with the online expert insights tracker.Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- AbbVie, Inc.
- Amgen, Inc.
- AstraZeneca Plc
- Bayer AG
- Bristol-Myers Squibb Company
- Eli Lilly and Company
- F. Hoffmann-La Roche Ltd.
- GSK Plc
- ICON Plc
- Insilico Medicine
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 172 |
| Published | May 2026 |
| Forecast Period | 2025 - 2032 |
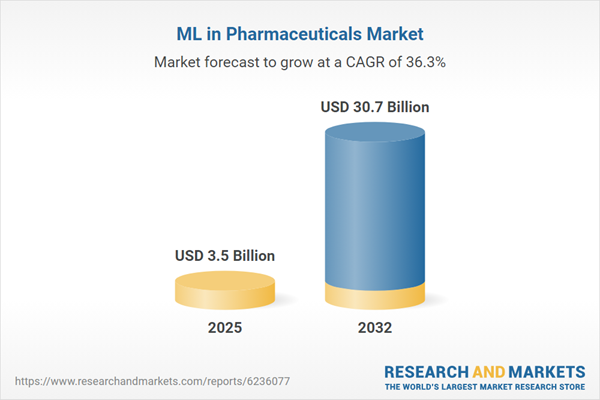
| Estimated Market Value ( USD | $ 3.5 Billion |
| Forecasted Market Value ( USD | $ 30.7 Billion |
| Compound Annual Growth Rate | 36.3% |
| Regions Covered | Global |