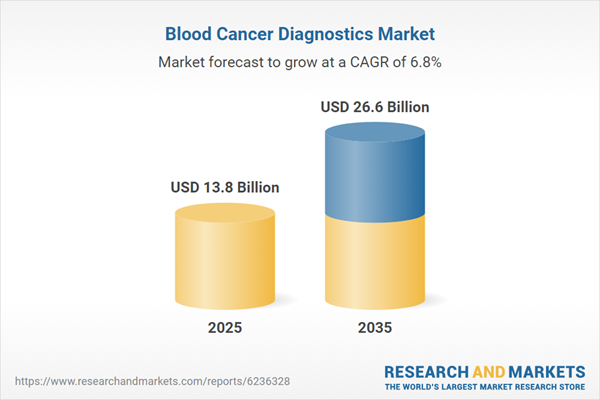
Market growth is driven by the increasing global incidence of hematological malignancies, supported by demographic shifts and improvements in disease detection capabilities. The growing need for early and accurate diagnosis is encouraging healthcare systems to prioritize advanced diagnostic solutions that enable timely intervention and improved patient outcomes. Rising awareness regarding cancer screening and the importance of early detection is further increasing the volume of diagnostic testing. Technological advancements in diagnostic platforms are significantly enhancing precision, sensitivity, and turnaround time, enabling better disease characterization and personalized treatment planning. The integration of advanced tools such as molecular diagnostics and sequencing technologies is transforming diagnostic workflows and improving clinical decision-making. In addition, expanding adoption of routine testing protocols is ensuring consistent demand across healthcare settings. Increasing investments in research and development, combined with the need for more efficient diagnostic approaches, continue to strengthen the overall growth trajectory of the blood cancer diagnostics market.
The assay kits and reagents segment generated USD 8.4 billion in 2025. This segment represents a critical component of the diagnostic ecosystem, supporting the detection and measurement of disease-related biomarkers. These products are widely utilized across multiple diagnostic methodologies and are essential for both clinical and research applications. Their standardized design, compatibility with laboratory systems, and ready-to-use formats contribute to operational efficiency and consistent testing outcomes. The ongoing demand for accurate and reproducible diagnostic results continues to drive adoption within this segment.
The blood tests segment accounted for USD 5.3 billion in 2025. This segment plays a foundational role in the diagnostic pathway by enabling the initial assessment of abnormalities through routine analysis of blood parameters. It serves as a primary step in identifying potential health concerns and guiding further diagnostic procedures. The widespread use of blood testing across healthcare facilities contributes to high testing volumes and consistent utilization. Its integration into standard clinical practices ensures ongoing demand for screening, monitoring, and evaluation purposes.
North America Blood Cancer Diagnostics Market accounted for 46.5% share in 2025. The blood cancer diagnostics market in the region is witnessing strong growth momentum, supported by the increasing burden of hematological malignancies. The rising occurrence of conditions such as leukemia, lymphoma, and multiple myeloma is significantly contributing to the demand for diagnostic solutions across North America. Demographic trends, including an aging population and evolving lifestyle-related risk factors, are further driving higher diagnosis rates. Growing patient volumes are increasing the urgency for timely and precise detection, leading to greater adoption of advanced diagnostic technologies across healthcare facilities and specialized laboratories. Additionally, the region benefits from well-established healthcare systems and high healthcare expenditure per capita, enabling broader access to advanced diagnostic capabilities.
Key players operating in the Blood Cancer Diagnostics Market include F. Hoffmann La Roche Ltd., Abbott Laboratories, Thermo Fisher Scientific Inc., Danaher Corporation, Becton Dickinson and Company (BD), Agilent Technologies, Inc., QIAGEN N.V., Bio Rad Laboratories, Inc., Sysmex Corporation, GE Healthcare, Guardant Health, Inc., Adaptive Biotechnologies Corporation, Invivoscribe Technologies, Inc., Precipio Diagnostics, and Exact Sciences Corporation. Companies in the Blood Cancer Diagnostics Market are focusing on innovation, strategic partnerships, and portfolio expansion to strengthen their competitive position. Significant investments are being made in research and development to enhance diagnostic accuracy and introduce advanced technologies. Collaborations with healthcare providers and research institutions are helping companies expand their clinical capabilities and improve product adoption. Firms are also working on expanding their global presence through geographic diversification and distribution network enhancements. Regulatory compliance and quality assurance remain key priorities to ensure product reliability.
Comprehensive Market Analysis and Forecast
- Industry trends, key growth drivers, challenges, future opportunities, and regulatory landscape
- Competitive landscape with Porter’s Five Forces and PESTEL analysis
- Market size, segmentation, and regional forecasts
- In-depth company profiles, business strategies, financial insights, and SWOT analysis
This product will be delivered within 2-4 business days.
Table of Contents
Companies Mentioned
The companies profiled in this Blood Cancer Diagnostics market report include:- Abbott Laboratories
- Adaptive Biotechnologies Corporation
- Agilent Technologies, Inc.
- Beckman Coulter, Inc.
- Becton, Dickinson and Company (BD)
- Bio Rad Laboratories, Inc.
- F. Hoffmann-La Roche Ltd.
- GE Healthcare
- Guardant Health, Inc.
- Invivoscribe Technologies, Inc.
- Precipio Diagnostics
- QIAGEN N.V.
- Sysmex Corporation
- Thermo Fisher Scientific Inc
- Exact Sciences Corporation
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 155 |
| Published | April 2026 |
| Forecast Period | 2025 - 2035 |
| Estimated Market Value ( USD | $ 13.8 Billion |
| Forecasted Market Value ( USD | $ 26.6 Billion |
| Compound Annual Growth Rate | 6.8% |
| Regions Covered | Global |
| No. of Companies Mentioned | 16 |