The research community and the biopharmaceutical industry have mobilized with unprecedented speed against the COVID-19 pandemic. Hence, organ-on-chip was founded to be one of the most promising and go-to technology during the development of treatment and prevention options that were then under clinical studies for the SARS-CoV-2 infection. For instance, in March 2022, using a novel organ-on-a-chip platform, a research team from the University of Toronto's Faculty of Applied Science & Engineering identified a molecule (QHREDGS) with the potential to combat one of the most severe complications of COVID-19 infections. Thus the new ongoing research studies of organ-on-chip for the treatment of COVID-19 are expected to have a positive impact on the market over the coming years.
An organ-on-a-chip is a microfluidic cell culture device that contains continuously perfused chambers. This chip develops a narrow channel for the blood and airflow in organs, such as the lung, gut, liver, heart, and other organs. Such devices produce multiple levels of tissue and organ functionalities, which are not feasible using conventional 2D and 3D culture systems. It offers a wide range of applications, such as disease modeling, patient stratification, and phenotypic screening. The organ-on-chip market is driven by factors such as a requirement for alternatives for animal testing and the need for early detection of drug toxicity along with new product launches and advancements in technology that are also responsible for driving the market. For instance, in December 2021, Emulate, Inc., stated that its Brain Chip was sent to the ISS National Lab to study the effects of microgravity on human brain physiology as part of the tissue-chips in space initiative sponsored by the NCATS at NIH and the ISS-NL.
Furthermore, in September 2021, Fidia Farmaceutici S.p.A. and BiomimX Srl signed a research collaboration agreement aimed at introducing and applying new Organs-on-Chip-based technological solutions to the intra-articular medical device discovery process. Moreover, in November 2021, AIM Biotech launched the idenTx 40 Plate, a high-throughput organ-on-a-chip system that let researchers at biotechnology and pharmaceutical companies faithfully recreate the function of human organs and tissues without using animals for research. The demand for personalized medication and the vast applications of organ-on-chip beyond the pharmaceutical industry are the major factors creating growth opportunities for market players.
However, the complexity of organ-on-chip models may hinder market growth over the forecast period.
Organ-on-Chips Market Trends
Lung-related Application is Expected to Exhibit a Significant Growth Over the Forecast Period
The lung-on-chip technology is a complex, three-dimensional model of a living, breathing human lung on a microchip. This device, made up of human lung and blood vessel cells, can predict the absorption of airborne nanoparticles and mimic the inflammatory response triggered by microbial pathogens. The device can also be used to test the effects of environmental toxins, the absorption of aerosolized therapeutics, and the safety and efficacy of new drugs. With the combination of microfabrication techniques with modern tissue engineering, lung-on-chip also offers a new in-vitro approach to drug screening by mimicking the complicated mechanical and biochemical behaviors of a human lung.The global COVID-19 pandemic caused by the novel coronavirus, SARS-CoV-2, necessitates rapid advances in research and development activities for different approaches to treating the infection. For instance, in September 2022, an airway-on-a-chip that mimics respiratory organs was developed by the CiRA research team using vascular endothelial cells and airway epithelial cells. With the device, they found that SARS-CoV-2 disrupts the vascular endothelial barrier.
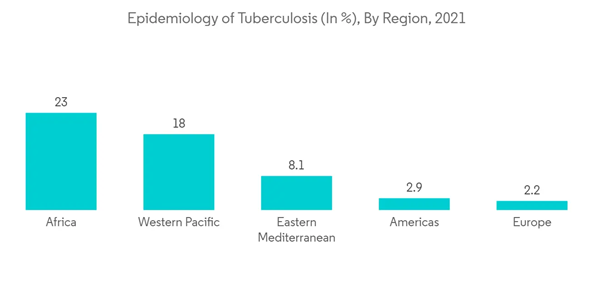
Furthermore, the development of new solutions to treat pulmonary diseases is of primary importance, as pulmonary diseases are found to be the most common conditions globally. According to the WHO, Global Tuberculosis Report 2022, it was estimated a global total of 10.6 million people fell ill with tuberculosis (TB) in 2021 which is equivalent to 134 cases per 100,000 population. The incidence rate of TB is also predicted to have grown by 3.6% between 2020 and 2021. Hence, the rise in the incidence of pulmonary diseases like tuberculosis increases the demand for the management of diseases using organ-on-chip technology thereby driving the growth of the market over the study period.
Hence, despite having already proven its worth, the lung-on-chip technology may also need to be engineered further, perhaps with muscle cells, to help test the effects of any treatment on the lungs, which may result in further growth of the market.
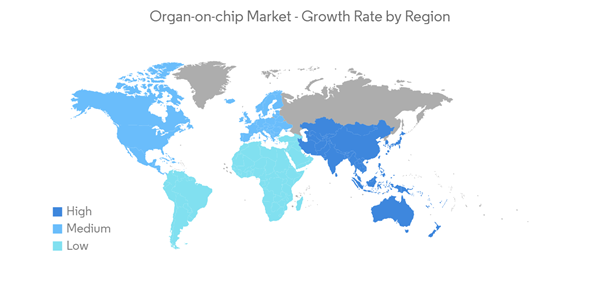
North America is Expected to Exhibit a Largest Market Share Over the Forecast Period
North America is anticipated to dominate the market over the period. The United States has the greatest market share in the North American area, because of rising R&D spending on drug discovery and biotechnology. It dominates the organ-on-chip market, owing to factors such as the availability of a wide range of services offered by major players which include customized designing of new chips with specific organ arrangements and an increase in toxicological testing of chemicals on the different types of organ cells. Huge investments are being made by public and private institutes for their research.For instance, as per the NIH, the estimated research funding for biotechnology in the United States is USD 7,847 million in 2021, and USD 8,142 million in 2022. This shows a gradual increase in research funding for biotechnology that helps in the research using organ-on-chip and drives the market over the forecast period.
Furthermore, several initiatives in providing organ-on-chip services along with approvals and launches in North America help the market to grow. For instance, in May 2022, Nikon Instruments Inc, in the United States stated that it is going to start organ-on-a-chip imaging and analysis contract services in an optimum manner for research and development for drug discovery. Similarly, in March 2021, AIM Biotech launched a contract research service, which uses its organ-on-a-chip technology to accelerate and improve drug discovery and development. These services further increase the scope of organ-on-a-chip imaging thereby boosting the growth of the market.
Thus, all aforementioned factors are expected to have a significant impact on the growth of the market in this segment.
Organ-on-Chips Industry Overview
The organ-on-chip market is moderately consolidated and consists of several major players. In terms of market share, a few of the major players currently dominate the market. The presence of major market players, such as Altis Biosystems, AxoSim, BiomimX SRL, Elveflow, Emulate Inc., InSphero, MIMETAS BV, Nortis Inc., Valo Health (Tara Biosystems Inc.), and TissUse GmbH, is in turn, increasing the overall competitive rivalry in the market.Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
This product will be delivered within 2 business days.
Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- Altis Biosystems
- AxoSim
- Bi/ond
- BiomimX SRL
- Elveflow
- Emulate Inc.
- Hesperos
- Allevi Inc.
- InSphero
- MIMETAS BV
- Netri
- Nortis Inc.
- Valo Health (Tara Biosystems Inc.)
- TissUse GmbH