The COVID-19 outbreak substantially impacted the mononucleosis diagnostics market as the pandemic increased the focus on the management of infectious diseases to limit their spread. This was attributed to the fact that COVID-19 was associated with Epstein-Barr virus (EBV) infections. For instance, as per an article published in September 2021 by BMC Infectious Diseases, out of the 128 patients with COVID-19, 13.3% were infected with Epstein-Barr virus reactivation. As per an update published in February 2022 by the University Health Network, long COVID-19 symptoms were also found in active EBV infections, and close to 70% of the COVID patients had reactivated EBV. It went on to say that EBV reactivation could be the cause of long COVID-19. Thus, such studies and updates indicate the demand for diagnosing EBV among the patient population increased during the pandemic. In addition, the market growth is stabilizing in the current scenario after COVID-19 as the worldwide restrictions have eased and the disease screening services have been resumed.
The major factors responsible for the growth of the global mononucleosis diagnostics market include rising health awareness and knowledge among patients, technological advancements leading to faster diagnosis, and the rise in the adolescent population. During the next few years, these factors are likely to continue to help the market grow.
Technological advancements leading to faster diagnosis are primarily driving the market's growth. For instance, in April 2021, Roche launched the Elecsys EBV panel, which consists of Elecsys EBV IgM, Elecsys EBV VCA IgG, and Elecsys EBV EBNA IgG immunoassays, in countries accepting the CE Mark. Additionally, in January 2022, Quanterix Corporation reported that its Simoa technology had been proven to be significant in identifying a high prevalence of EBV associated with multiple sclerosis (MS). Thus, growing technological advances in the mononucleosis diagnostics market are expected to increase demand over the forecast period.
Moreover, as per the study published in August 2022 by Frontiers in Immunology, within 10 years from the index date, the incidence of MS was 22.6 cases per 100,000 person-years among patients with infectious mononucleosis but only 11.9 cases per 100,000 person-years among individuals without infectious mononucleosis. So, the growing number of cases of mononucleosis is likely to help the study market grow over the next few years.
Still, the fact that there aren't many tests for diagnosing mononucleosis is a big reason why the market isn't growing faster.
Mononucleosis Diagnostic Market Trends
The Monospot Test Segment by Test Type is Expected to Hold the Significant Market Share During the Forecast Period
By test type, the monospot test segment is expected to witness significant growth. The monospot test is a latex agglutination test that utilizes equine erythrocytes as the primary substrate and tests for specific heterophile antibodies produced by the human immune system in response to EBV infection. This test is expected to hold the largest market share, owing to the benefits associated with it, such as non-interference with other tests, accurate results, rapid detection of antibodies, and cost-effectiveness. Furthermore, an article published in StatPearls in August 2021 stated that mononucleosis is diagnosed using the monospot (or heterophile antibody) test, which is nearly 100% specific for the disease. This test's sensitivity is closer to 85 percent. Such studies greatly increase the acceptance of monospot test types among the patient population, thereby contributing to the segment's growth.According to the study published in the Clinical Journal of Sports Medicine in November 2021, the monospot test had a sensitivity and specificity of 80.0% and 90.6%, respectively. Increased transaminase levels have a strong correlation with acute infectious mononucleosis and may aid in the diagnosis. A positive monospot test result is the most reliable way to diagnose infectious mononucleosis (IM). Thus, high sensitivity and specificity are likely to help the growth of the segment over the next few years.
However, as a primary Epstein-Barr virus infection symptom, infectious mononucleosis in adults is uncommon. The teen population, which is more prone to these illnesses, is anticipated to drive market expansion. Thus, the above-mentioned factors are expected to drive the growth of this segment over the forecast period.
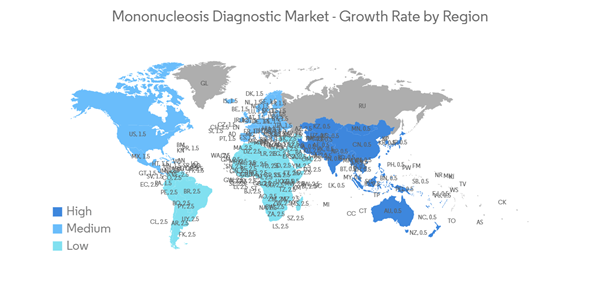
Asia-Pacific Accounted for the Highest Growth Rate in the Global Market
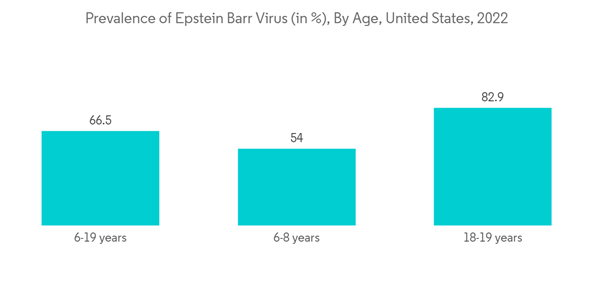
The Asia-Pacific region holds the highest growth rate for the mononucleosis diagnostic market, and it is expected to show a similar trend over the forecast period without significant changes. The rising adolescent population in the region susceptible to EBV infection is one of the major factors that is expected to drive the growth of the market over the forecast period. For instance, according to an article published by the Lancet in February 2022, mononucleosis is a disease that primarily affects adolescents and is caused by the Epstein-Barr virus. However, this virus is highly prevalent, with two maximum occurrence peaks in an individual's life: firstly, around 3-4 years of age, and then again at 15-16 years of age. When an Epstein-Barr virus develops in childhood, the risk of developing mononucleosis is quite low, but it is substantially higher in adolescence. This indicates that a large number of people are infected with the Epstein-Barr virus without being aware of the condition.Moreover, according to the United Nations Children's Fund, in January 2022, the East Asia and Pacific region will be home to 329 million adolescents, or about 25% of all teenagers worldwide. The high youth population indicates a significant demand for mononucleosis diagnostic tests. Moreover, as per an article published by Verywell Health in November 2022, in the United States, 25% to 50% of children living in lower socioeconomic circumstances will have been infected with EBV by age 4. Additionally, 75% of young adults will be diagnosed with mono after an EBV infection. This, along with the increasing cases of EBV followed by mononucleosis in the region, is expected to boost the growth of the market over the forecast period.
In addition, the presence of key companies such as Cardinal Health and Abbott in the region is also contributing to the market's growth. Since there are a lot of teenagers in the area, there are likely to be a lot of great market opportunities in the coming years.
Mononucleosis Diagnostic Market Competitor Analysis
The mononucleosis diagnostics market is dominated by both major and small players. However, in the niche segment of the market, there are only a few small market players. The major market players, such as Abbott Laboratories, Danaher Corporation, Thermo Fisher Scientific Inc., Cardinal Health Inc., and Bio-Rad Laboratories, are focusing on the development of advanced products in order to achieve faster detection.Additional benefits of purchasing the report:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
This product will be delivered within 2 business days.
Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- Abbott Laboratories
- Arlington Scientific Inc.
- Bio-Rad Laboratories Inc.
- Cardinal Health Inc.
- Immunostics Inc.
- Meridian Bioscience
- Sekisui Diagnostics
- Thermo Fisher Scientific Inc.
- Launch Diagnostics Limited
- Beckman Coulter Inc.
- Diasorin
- BioMérieux