During the COVID-19 outbreak, all outpatient treatments were postponed or restricted to reduce the risk of viral transmission, as most chronic therapies were deemed elective. As per the article published in the Springer Journal in September 2022, during the COVID-19 pandemic, the number of treatments for endometriosis patients was reduced, especially during the first lockdown (March 2020 to May 2020). There was a 37% reduction in the endometriosis outpatient department and a 90% reduction in surgical endometriosis. Thus, the decline in endometrial consultations and surgical procedures during COVID-19 significantly impacted market growth during the initial stages of the pandemic. However, owing to the resumption of endometrial surgeries after the relaxation of strict lockdown regulations during the post-pandemic period, the market is expected to grow over the forecast period.
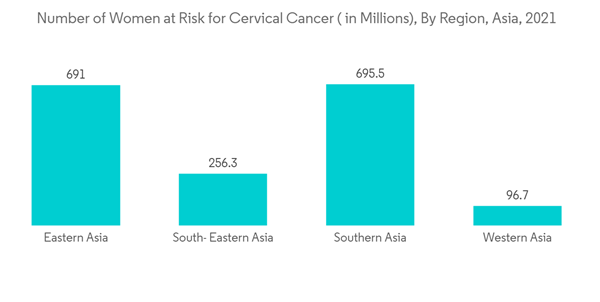
The major factor attributing to the growth of the market is the increase in the prevalence of gynecological disorders in women, like cervical cancer, menorrhagia, abnormal vaginal bleeding, and PCOD, or polycystic ovarian disease. According to the article published in Elsevier in June 2021, overall, 116 million women were affected by polycystic ovarian syndrome across the world. Additionally, in the study published in the HPV Information Center in October 2021, every year, 351,720 women were diagnosed with cervical cancer in Asia. The report also estimated that a population of 1766.41 million women aged 15 years and older were at risk of developing cervical cancer in Asia. Thus, the growing prevalence of various gynecological diseases is expected to drive demand for endometrial ablation devices owing to the effective treatment, thereby boosting market growth over the forecast period. The increasing number of product launches by various key market players in order to meet the growing demand for endometrial ablation devices is also expected to fuel the market's growth during the forecast period. For instance, in November 2021, Hologic, Inc., launched the NovaSure V5 global endometrial ablation (GEA) device at the 50th Global Congress of the American Association of Gynecologic Laparoscopists (AAGL) in the United States.
Thus, the above-mentioned factors, such as an increase in the prevalence of gynecological disorders and frequent product launches by various market players, are expected to contribute to the growth of the market. However, stringent regulatory policies are expected to impede the growth of the market over the forecast period.
Endometrial Ablation Devices Market Trends
Radiofrequency Endometrial Ablation Device is Expected to Hold Significant Share in the Market Over The Forecast Period
Radiofrequency ablation devices are used to transmit radio frequency energy and destroy the tissue lining the uterus (the endometrium). These devices ensure a reduced risk of infection. The notable share of radiofrequency endometrial ablation devices is accounted for by factors such as high safety and fast recovery, increasing awareness related to this technique in developing countries, and high preference from several medical practitioners. Apart from the advantages of the devices, the factors driving segment growth are the increasing prevalence of cervical cancer, menorrhagia, and other gynecologic disorders.According to the NCBI study published in August 2022, in the clinical trial conducted to test the safety and efficacy of the Cerene device, a radiofrequency endometrial ablation device, 91% of the enrolled subjects reported that they had no limitations in the activity of the device, and 85% of the subjects were satisfied with the performance of the device. Thus, the higher safety and efficacy of the radiofrequency ablation system are expected to increase segment growth over the forecast period. Moreover, the growing prevalence of endometrial cancer is expected to boost segment growth. As per the ACS 2022 update, there were more than 600,000 survivors of endometrial cancer in the United States. According to the same source, around 65,950 new cases of cancer of the body of the uterus (uterine body or corpus) are expected to be diagnosed in 2022. Thus, the growing prevalence of endometrial cancer is expected to increase demand for endometrial ablation devices.
Additionally, the increasing number of product approvals is also expected to contribute to the growth of the market. For instance, in September 2021, Gynesonics received approval from the US FDA for their next-generation Sonata System 2.2 radiofrequency ablation of fibroid tumors. This Sonata technology platform integrates a commercial intrauterine ultrasound system with an advanced radiofrequency ablation device to provide incision-free, uterus-preserving treatment. Thus, the high safety and efficiency of the radiofrequency endometrial ablation devices and the approval of the advanced products are expected to contribute to the growth of the studied segment.
North America is Expected to Hold Significant Share in the Market Over the Forecast Period
North America is expected to hold a significant share of the overall market throughout the forecast period. Escalating cases of gynecological disorders, rising healthcare infrastructure, and the strong establishment of major market players to offer innovative endometrial ablation devices are accountable for promoting the endometrial ablation devices market in this region. According to the NCBI study published in May 2022, endometriosis is a chronic inflammatory pain disorder that affects 6.5 million women in the United States each year. Additionally, the data published by the Canadian Cancer Society in 2022 mentioned that an estimated 1,450 Canadian women will be diagnosed with cervical cancer in 2022. Hence, the growing prevalence of endometritis and gynecological disorders in this region is expected to drive the demand for endometrial ablation devices, thereby contributing to the growth of the market in this region.Additionally, key market players' strategies, such as acquisitions and mergers, and rising investments support market growth. For instance, in October 2021, Hologic, Inc. acquired Bolder Surgical for approximately USD 160 million. The complementary acquisition of Bolder Surgical added to the Hologic surgical product line, which includes the Novasure endometrial ablation system for the treatment of abnormal uterine bleeding. Also, in February 2022, CooperSurgical agreed to spend USD 875 million on Cook Medical's reproductive health portfolio, which includes fertility, obstetrics, gynecology, and in vitro fertilization medical equipment.
Endometrial Ablation Devices Market Competitor Analysis
The endometrial ablation devices The market is moderately competitive and consists of several major players. Some of the companies that are currently dominating the market are Boston Scientific Corporation, Cooper Surgical, Medtronic, Johnson & Johnson, Olympus Corporation, Richard Wolf GmbH, Minerva Surgical, Inc., AngioDynamics, and Hologic, Inc.Additional benefits of purchasing the report:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
This product will be delivered within 2 business days.
Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- Boston Scientific Corporation
- Cooper Surgical
- Medtronic Plc
- Johnson & Johnson
- Olympus Corporation
- Richard Wolf GmbH
- Minerva Surgical, Inc.
- AngioDynamics
- Hologic, Inc.
- AEGEA Medica Inc
- Teoranta
- Veldana Medical SA