The COVID-19 outbreak has significantly impacted the NAAT market as it was mainly recommended for the detection of a potent SARS-CoV-2 infection was a nucleic acid amplification (NAAT) test that detects one or more specific RNA-specific strains of the virus. According to the study published in Clinical Microbiological Infection in March 2021, in the COVID-19 diagnostic, the overall NAAT sensitivity was 89.1% and the specificity was 98.9%. As a result of the exceptional sensitivity and specificity of NAAT testing in the diagnosis of COVID-19, the demand for such tests spiked amid the pandemic. Therefore, the market during the pandemic might have been significantly impacted due to rise in sales of the NAAT test. Furthermore, the market is expected to show significant growth post-pandemic owing to the emergence of multiple strains of the COVID-19 viruses and other infectious diseases.
The major factors for the growth of the nucleic acid amplification testing (NAAT) market include the rising prevalence of infectious diseases, increasing demand for advanced diagnostic measures, and rising investment in the development of new biotechnological diagnostic techniques.
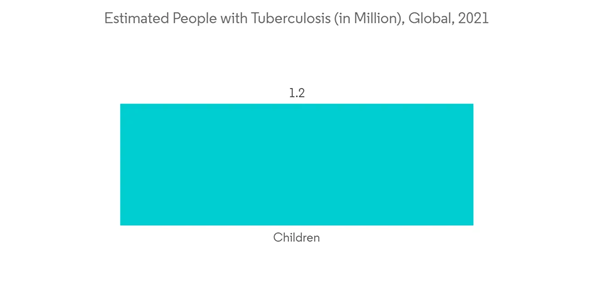
There is a high prevalence of infectious diseases such as tuberculosis and HIV, which are further responsible for the increasing demand for diagnostic tests, thereby positively impacting the growth of the studied market. For instance, as per the WHO in October 2022, an estimated 10.6 million people fell ill with tuberculosis(TB) worldwide in 2021. Among them 6 million are men, 3.4 million are women and 1.2 million are children. Thus, such a huge prevalence of infectious diseases such as tuberculosis is expected to propel the market's growth due to the rise in NAAT testing adoption.
Rising investments, adoption of key strategies, and product launches are further expected to drive the market in the future. For instance, in December 2021, Roche launched infectious disease tests on the Cobas 5800 System, a new molecular laboratory instrument, in countries accepting the CE mark. These include cobas HIV-1, cobas HBV, cobas HCV, cobas HIV-1/HIV-2 Qualitative, and the cobas omni-Utility Channel kit. Cobas HIV-1/HIV-2 Qualitative is an in vitro nucleic acid amplification test for the qualitative detection and differentiation of HIV-1 and HIV-2 in human serum, plasma, and dried blood spots. Such launch of innovative products for NAAT testing is expected to drive market growth due to increased adoption.
Thus, all the aforementioned factors such as rising prevalence of infectious diseases, rising initiatives of key market players in the launch of diagnostics for NAAT, and rising investments in new biotechnological diagnostic tests are expected to boost the market's growth over the forecast period. However, a lack of skilled professionals and higher costs associated with these tests may restrain the market's growth.
Key Market Trends
The Infectious Disease Segment is Expected to Hold a Significant Market Share Over the Forecast Period
Infectious diseases are caused by pathogenic microorganisms such as bacteria, viruses, parasites, or fungi. The diseases can spread, directly or indirectly, from one person to another. The common infectious diseases are strep throat and urinary tract infections (caused by bacteria), HIV/AIDS, the common cold (caused by a virus), influenza, STIs, tuberculosis, and athlete's foot (common fungus), among others.According to the World Health Organization in August 2022, sexually transmitted infections (STIs) have a significant impact on sexual and reproductive health worldwide and over 1 million STIs are acquired every day in the world. Furthermore, according to the same source, 374 million new infections are estimated with STIs such as chlamydia, gonorrhea, syphilis, and trichomoniasis every year in the world. Thus, such a high incidence of infectious diseases is expected to increase the demand for NAAT testing, driving the market growth over the forecast period.
Furthermore, according to the study published in Infectious Medicine in March 2022, the NAAT has higher sensitivity in the detection of tuberculosis compared to other diagnostic tests such as smear and culture tests. The high sensitivity of this test in the detection of infectious diseases such as tuberculosis is expected to drive segment growth due to its increased adoption in tuberculosis testing.
Thus, the rising prevalence of various infectious diseases and the rising importance of NAAT in the diagnosis of infectious diseases is expected to boost the segment's growth over the forecast period.
North America is Expected to Hold a Significant Market Share Over the Forecast Period
The North American region is expected to dominate the global market owing to the presence of better healthcare infrastructure, the presence of prominent market players, and the rising prevalence of infectious and chronic diseases.For instance, according to the ACS Cancer Facts & Figures 2022, around 1,918,030 new cancer cases are estimated to be reported in the United States in the year 2022. Furthermore, according to Statistics Canada in June 2022, it is estimated that around 233,900 people in Canada will be diagnosed with cancer in 2022. The huge cancer burden is increasing the need for diagnostic tests like NAAT driving the market growth in the region.
Additionally, a few of the key market players in the country are developing advanced diagnostics kits to compete with the existing products, while others are acquiring and partnering with the other companies trending in the market. For instance, in September 2022, BD and CerTest Biotec developed a molecular polymerase chain reaction (PCR) assay for the monkeypox virus. Such developments are further bolstering the market growth in the region due to the increased adoption of NAAT testing.
Furthermore, in November 2022, Pulse Scientific was named the national distributor in Canada for COVID-19 & Flu at-home molecular tests developed by Lucira Health. The Lucira testing platform is a Nucleic Acid Amplification Test (NAAT) with sensitivity and specificity comparable to lab-based PCR assays. Therefore, the rising distribution of NAAT in Canada is expected to drive market growth in the region due to the rise in adoption by people for testing infectious diseases conveniently at home.
Thus, the above-mentioned factors such as the rising prevalence of cancers, and rising initiatives from the key market players in launching their products are expected to witness high growth.
Competitive Landscape
The nucleic acid amplification testing (NAAT) market is moderately fragmented. The factors owing to the competition include rising infectious diseases, the rising launch of products, and rising partnerships between key players, among others. Some of the market players are Abbott Laboratories, Becton, Dickinson and Company, bioMérieux SA, Bio-Rad Laboratories Inc., Grifols SA, Hoffmann-La Roche Ltd., Hologic Inc., and Illumina IncAdditional benefits of purchasing the report:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
This product will be delivered within 2 business days.
Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- Abbott Laboratories
- Becton, Dickinson and Company
- bioMérieux SA
- Bio-Rad Laboratories Inc.
- Grifols SA
- Hoffmann-La Roche Ltd
- Hologic Inc.
- Illumina Inc.
- Siemens Healthcare GmbH
- Beckman Coulter Inc. (Danaher Corporation)
- Tecan Trading AG
- Seegene Inc