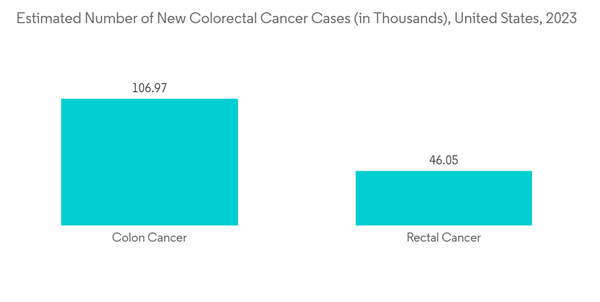The GI Stool Testing Market size is estimated at USD 507.52 million in 2024, and is expected to reach USD 696.67 million by 2029, growing at a CAGR of 6.54% during the forecast period (2024-2029).
This product will be delivered within 2 business days.
Key Highlights
- COVID-19 affected the GI stool testing market significantly. Various clinical studies were conducted to confirm the presence of coronavirus among the population. For instance, an article published in Frontiers in Medicine in May 2021 showed that Cepheid Xpert Xpress SARS-CoV-2 (tests with three gene targets (E, N2, and RdRP) assay was used for the accurate identification of viral RNA in stool samples. In addition, the Xpert Xpress SARS-CoV-2 test uses the GeneXpert benchtop equipment to enable qualitative detection of the virus in about 45 minutes.
- The inclusion of stool PCR in the testing algorithm was anticipated to increase clinical sensitivity. Furthermore, as per the same source, the detection of viral RNA in stool was confirmed after 33 days, and stool PCR confirmed the diagnosis of SARS-CoV-2 in two individuals whose upper respiratory tract PCR results were negative. This has increased the utilization of stool testing techniques for confirming COVID-19 infection among the population, which is further anticipated to fuel the market growth over the forecast period. However, currently, as COVID-19 cases have been subsidized, the rising application of Gi stool testing leads to the stable growth of the market over the coming years.
- The factors such as the growing burden of gastrointestinal diseases and rising preference for point-of-care tests are boosting the market growth. For instance, a 2022 update by Bower Cancer UK showed that bowel cancer is the 4th most common cancer, and approximately 43,000 people are diagnosed with bowel cancer every year in the United Kingdom. This is expected to increase the demand for stool DNA testing, which is used to screen for colon cancer in people with no symptoms, hence propelling the market growth.
- Additionally, as per an article published in PLOS One in April 2021, it has been observed that the overall prevalence of hemorrhoids was higher in the male population in Northwest Ethiopia. The risk of having hemorrhoids was shown to be higher in people who were overweight and had constipation problems. Thus, the high prevalence of hemorrhoids among the population is anticipated to increase the need for a fecal occult blood test to early detect the disease and hence boost the market growth.
- Furthermore, the rising product launches increase the availability of stool testing kits and products in the market, which in turn is anticipated to fuel the market growth. For instance, in June 2022, Prenetics Group Limited, one of the global leaders in genomic and diagnostic testing, launched a novel, non-invasive, at-home screening test, ColoClear by Circle (ColoClear), for detecting early signs of colorectal cancer in Hong Kong.
- Therefore, owing to the factors such as the high burden of gastric cancer and other related diseases, the studied market is expected to grow over the forecast period. However, the high cost of instruments is expected to impede the growth of the GI stool testing market over the forecast period.
GI Stool Testing Market Trends
Reagent Segment is Expected to Witness Significant Growth in the GI Stool Testing Market Over the Forecast Period
- The reagent segment is expected to witness significant growth in the GI stool testing market over the forecast period owing to the factors such as the rising prevalence of gastrointestinal diseases such as irritable bowel syndrome, GERD, and others, as well as rising demand for early detection of the diseases.
- Reagents are substances that are often used for examining and analyzing. Reagents are expected to dominate the GI stool testing market with developing diagnostic technology. Chemiluminant assays are used as a diagnostic aid for the detection of gastrointestinal infections. It uses enzyme-labeled antibodies for detection, which is known to give an accurate diagnosis.
- According to an article published in Microorganisms in June 2022, it was observed that microscopic examination of feces is still a common method for diagnosing intestinal parasite infections, which necessitates the use of reliable reagents and qualified personnel. In addition, as per the same source, the ready-to-use concentration methods for detecting parasite eggs in the ParaFlo tests (Eurobio Ingen) are expected to improve reagent traceability and manipulation ease. This is anticipated to boost the demand for reagents for detecting various gastrointestinal diseases, hence propelling segment growth.
- Furthermore, stool testing is also beneficial for the early diagnosis of colon cancer, which is also expected to propel segment growth. For instance, as per an article published in the Clinical and Translational Gastroenterology in April 2022, the multitarget stool DNA test with fecal immunochemical test (sDNA-FIT) is recommended as a potential approach for colorectal cancer screening, in people aged 45 years and above and for those who are at average risk, in all significant United States guidelines. The sDNA-FIT analyzes 11 biomarkers, including human hemoglobin, NDRG4, and BMP3 aberrant methylation, KRAS point mutations, NDRG4 and BMP3 point mutations, and human hemoglobin. Hence the adoption of stool testing for various other GI-related diseases is anticipated to boost the market growth.
- Moreover, in June 2022, Meridian Bioscience, Inc. launched two new sample-specific master mixes, Lyo-Ready Direct DNA qPCR Stool Mix and Lyo-Ready Direct RNA/DNA qPCR Stool Mix. These innovative master mixes improve the molecular detection of DNA and RNA from crude fecal specimens while allowing room temperature stabilization of diagnostic assay.
- Therefore, owing to the factors such as the rising research studies and increasing adoption of reagents in stool testing, the studied segment is expected to grow over the forecast period.
North America is Expected to Hold a Significant Share in the Market and is Expected to Witness the Same Over the Forecast Period
- North America is expected to hold a significant share of the market and is expected to witness the same over the forecast period owing to the factors such as a higher incidence te of gastrointestinal diseases like colorectal cancer. For instance, according to 2023 statistics published by the ACS, about 106,970 new cases of colon cancer and 46,050 new cases of rectal cancer cases are expected to be diagnosed in the United States in 2023.
- Also, as per an article published in the National Library of Medicine (NLM) in July 2022, gastroesophageal reflux disease (GERD) is one of the most widespread gastrointestinal conditions, affecting 20% of adults in Western countries such as the United States. In addition, the estimated prevalence of GERD in the US population ranges from 18.1% to 27.8%. Furthermore, per the data published by the Canadian Cancer Society, in May 2022, colorectal cancer is expected to be the fourth most diagnosed cancer in Canada. Nearly 24,300 Canadians were diagnosed with colorectal cancer, which represented 10% of all new cancer cases in Canada in 2022.
- Furthermore, the rising company activities in developing innovative test kits and increasing product launches are also contributing to the market growth. For instance, in January 2023, Geneoscopy submitted a premarket approval application to FDA for its stool-based colorectal cancer screening test. Such initiatives may lead to the introduction of new products in the market in the near future and thus drives the growth of the market.
- Therefore, owing to the factors such as the rising burden of colon cancer and GERD among the population and increasing product launches, the studied market is anticipated to augment the market growth over the forecast period.
GI Stool Testing Industry Overview
The GI stool testing market is moderately fragmented in nature, with the presence of major players in the market. Collaborations, mergers, and acquisitions by companies related to GI stool testing are expected to boost market growth. Some of the companies which are currently dominating the GI stool testing market are bioMérieux SA, DiaSorin S.p.A, Cenegenics Corporation, Abbott Laboratories, Epitope Diagnostics Inc., Genova Diagnostics, Danaher Corporation (Beckman Coulter, Inc.), ScheBo Biotech AG, CTKBiotech Inc. Inc., and Cardinal Health.Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
This product will be delivered within 2 business days.
Table of Contents
1 INTRODUCTION
4 MARKET DYNAMICS
5 MARKET SEGMENTATION (Market Size by Value - USD Million)
6 COMPETITIVE LANDSCAPE