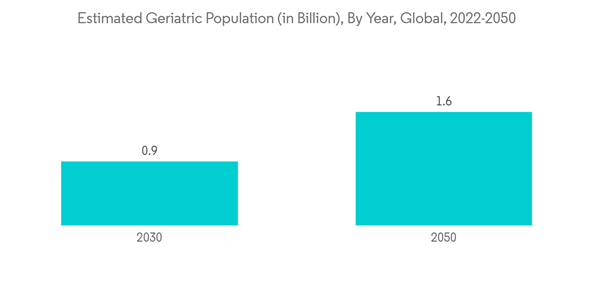The COVID-19 pandemic has impacted the global healthcare system, including the inhaled nitric oxide market. For instance, an article published in the journal IJP in May 2021 reported that in patients with COVID-19 with acute respiratory distress syndrome (ARDS), nitric oxide was linked to a slight increase in oxygenation. Initially, the market witnessed significant growth due to the rising number of COVID-19 cases, and the significant effect of liquid nitric oxide was linked to overcoming ARDS in COVID-19 patients by increasing arterial oxygenation. Now that COVID-19 cases have declined, it is anticipated that the demand for inhaled nitric oxide may stabilize.
The factors driving the growth of this market are the rising prevalence of respiratory and other associated diseases and ongoing research and development activities. The most prevalent element driving this market is rising cases of chronic respiratory diseases such as asthma, chronic obstructive pulmonary disease (COPD), acute respiratory disease syndrome (ARDS), and tuberculosis (TB), among others.
Asthma and COPD are considered the most common chronic disease worldwide and this has driven the need for inhaled nitric oxide. For instance, in August 2022, Lung India reported that the prevalence of Asthma among Indian children was around 18.2%. The same source suggests that there is a high prevalence of severe asthma in the emerging markets of the Middle East and Africa as well as Latin America, compared to the mid-range prevalence in North America and Europe.
Similarly, the rising cases of malaria reported globally are increasing the demand for inhaled nitric oxide. The inhaled nitric oxide reduces endothelial activation and parasite accumulation in the brain and enhances survival in cerebral malaria cases. In July 2022, WHO reported that in the year 2020, 241 million cases of malaria were estimated globally.
The ongoing research and development activities and clinical trials being conducted for the development of new drugs to prevent this disease are also expected to fuel the market growth. For instance, in February 2022, Glenmark launched a nitric oxide nasal spray (FabiSpray) in India for the treatment of adult patients with COVID-19 in partnership with SaNOtize.
While these factors are poised to give the market an impetus, strict application norms across various regions may slow down its growth over the studied period.
Inhaled Nitric Oxide Market Trends
The Asthma and COPD Segment is Expected to Witness a Significant Growth Over the Forecast Period.
Asthma and COPD are considered among the most common respiratory diseases affecting the population and life-threatening conditions affecting patients' regular breathing. Asthma is a persistent condition that stretches and narrows the air passages of the lungs, causing constant assaults of breathlessness, bronchospasm, and reversible obstruction of airflow.A large rise in the consumption of cigarettes contributes to asthma and COPD symptoms. In addition, the advancement in industrialization has also contributed to an increase in the number of cases of asthma and COPD, which, over the forecast period, may drive the demand for inhaled nitric oxide.
An article published in the journal JGH in October 2021 reported that an estimated 300 to 400 million people globally live with chronic obstructive pulmonary disease. The growing burden of COPD particularly concerns low- and middle-income countries (LMICs) due to increased smoking rates, household- and ambient air pollution, and other exposures, coupled with large and aging populations. Thus, the increasing number of COPD and asthma cases and the high dependence of the prevalent population on nitric oxide to overcome the symptoms are expected to drive the growth of this segment.
Moreover, an increasing geriatric population globally is boosting the growth of this segment. For instance, the United Nations reported that in the year 2022, there were 771 million people aged 65 years or over globally. The older population is projected to reach 994 million by 2030 and 1.6 billion by 2050. Since asthma is a widely prevalent disease found in people over the age of 65, the growing geriatric population in the segment is expected to drive its growth.
Similarly, a report published by the Global Initiative for Chronic Obstructive Lung Disease in 2022, stated that the prevalence and burden of COPD are projected to increase shortly due to continued exposure to COPD risk factors, a rise in the incidence of interstitial lung diseases, and aging population across the world. This will likely boost the demand and market for inhaled nitric oxide.
North America Region is Expected to Witness a Significant Growth Over the Forecast Period.
North America is expected to witness significant growth over the forecast period. This growth can be attributed to the rising incidence of diseases such as COPD and asthma, the aging population, increasing research and development activities, and the presence of key market players.There has been a rise in the prevalence of disorders such as neonatal respiratory disorder, tuberculosis, and ARDS. For instance, in March 2022, the Government of Canada reported 4.7 active TB cases per 100,000 persons. The corresponding rate for females was 4.2 cases per 100,000, compared to 5.1 cases per 100,000 for men. Similarly, an article published by Scientific Reports in August 2022 reported that in Canada, 3.8 million people were living with asthma during the COVID-19 pandemic. Thus, high cases of respiratory disorders are increasing the demand for inhaled nitric oxide, thereby driving the studied market.
Similarly, as per the April 2021 statistics by the Asthma and Allergy Foundation of America, about 20 million adults aged 18 years or above in the United States have asthma. Thus, the prevalence of such disorders in the country indicates a consistent demand for inhaled nitric oxide, thereby contributing to the growth of the market studied in the United States.
Also, new product launches, mergers, acquisition, and other development is boosting the growth of liquid nitric oxide in the studied region. For instance, in September 2022, Mallinckrodt PLC promulgated the submission of a 510(k) premarket notification application to the USFDA for an investigational inhaled nitric oxide delivery system for INOmax (nitric oxide) gas, for inhalation. INOmax (nitric oxide) gas, for inhalation, is an approved treatment to improve oxygenation and reduce the need for extracorporeal membrane oxygenation in term and near-term (34 weeks gestation), neonates, with hypoxic respiratory failure associated with clinical or echocardiographic evidence of pulmonary hypertension in conjunction with ventilatory support and other appropriate agents.
Furthermore, the rise in the number of initiatives undertaken by government and non-government organizations is increasing the overall revenue. For instance, in 2021 CDC’s NACP provided funds for educating asthma-affected patients. Such initiatives are likely to increase awareness about asthma, which will increase the adoption of inhaled nitric oxide in overcoming the symptoms of asthma, thereby driving the growth of the studied market in the region.
Inhaled Nitric Oxide Market Competitor Analysis
The inhaled nitric oxide market is competitive due to the presence of many companies operating globally and regionally. The competitive landscape includes an analysis of a few international as well as local companies which hold market shares and are well-known, such as Air Liquide Healthcare, Matheson Tri-Gas Inc., Merck KGaA, Mallinckrodt Pharmaceuticals (Novoteris), BOC Healthcare, Nu-Med Plus Inc., Perma Pure LLC, Praxair Distribution Inc., HALMA PLC, LINDE PLC, and Bellerophan Therapeutics Inc.Additional benefits of purchasing the report:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
This product will be delivered within 2 business days.
Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- Air Liquide Healthcare
- BOC Healthcare
- Matheson Tri-Gas Inc.
- Merck KGaA
- Mallinckrodt Pharmaceuticals (Novoteris)
- Nu-Med Plus Inc.
- Perma Pure LLC
- Praxair Distribution Inc.
- HALMA PLC
- LINDE PLC
- Bellerophan Therapeutics Inc.