Key Highlights
- COVID-19 impacted market growth slightly due to the cancellations of elective procedures involving hospitalization other than COVID-19. However, the market growth recovered in the last few years since the restrictions were lifted. The new variants of COVID-19 have no significant impact on the studied market in the current scenario. Hence, the market is expected to register a stable growth rate during the forecast period.
- The advantages of intraosseous infusion (IO) over other routes of administration of drugs are expected to propel market growth. For instance, a research article published in the European Journal of Trauma and Emergency Surgery in February 2021 included nine studies comprising 1,218 trauma patients and 1,432 device insertions. The insertion success rate was 95%, and the incidence of complications was 0.9%. The article stated that intraosseous catheters have high insertion success rates and a low incidence of complications in trauma patients.
- Additionally, an article published in February 2022 by the National Center for Biotechnology Information (NCBI) stated that intraosseous infusion is an effective and safe technique, that increases patient survival. As a result, it is critical that all nursing professionals understand how to use the various intraosseous devices in situations where immediate peripheral venous access is not possible. The advantages and rising awareness about IO infusion are expected to propel the demand for IO infusion kits during the forecast period.
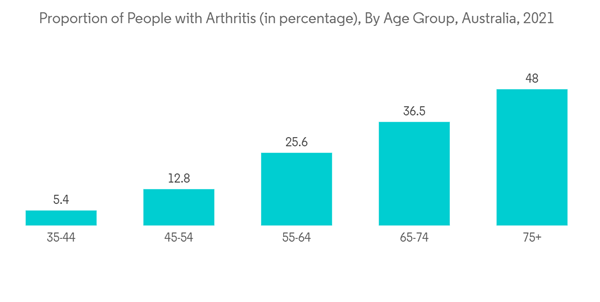
- Furthermore, the rising burden of arthritis and trauma is expected to propel the demand for IO infusion as these diseases are associated with difficulty in establishing venous access, which is made possible by the use of intraosseous needles. For instance, as per the Australian Bureau of Statistics 2023 update, one in eight (12.5%) people of all ages had arthritis in Australia in 2021. Additionally, as per versus arthritis 2022 report, 20.3 million people had a musculoskeletal (MSK) condition such as arthritis or back pain in the United Kingdom in 2021. Therefore, the high prevalence of arthritis worldwide is expected to propel the demand for IO infusion for the greater bioavailability of drugs for the treatment of diseases, thereby augmenting market growth.
- Therefore, owing to the aforementioned factors, such as the advantages of IO infusion and the high burden of arthritis worldwide, the studied market is anticipated to witness growth over the analysis period. However, the high cost associated with Procedures and stringent regulatory approvals and product recalls is expected to restrain the market growth.
Intraosseous Infusion Kits Market Trends
Hospital Segment is Expected to Hold a Significant Market Share Over The Forecast Period
- Hospitals are the primary care settings for treating medical conditions, including arthritis, trauma, and other bone disorders. The hospital segment is expected to witness a significant growth rate during the forecast period owing to the rising number of injured and traumatic patients admitted to hospitals that require IO infusion kits as a first line of treatment for immediate relief.
- The high number of hospitalizations for trauma and burn patients boosts the demand for IO infusion kits as such people have compromised intravenous access and need immediate delivery of life-saving fluids and medications. For instance, research published in April 2021 in the Saudi Journal of Biological Science conducted at the King Khalid Hospital Prince Sultan Center for Health Services and Prince Sattam Bin Abdulaziz University Hospital in the Kingdom of Saudi Arabia surveyed patients with burns. The study stated that the prevalence of first-degree burns was 12.8%, second-degree burns were 71.1%, and third-degree burns were 16.1%. Therefore, the high number of burns worldwide is expected to propel the demand for hospitalization, which is further expected to boost market growth as it enables the quick infusion of intravenous fluids and drugs into the systemic venous system, which cannot be achieved via another route of administration.
- Furthermore, in July 2022, a press release by ISTAT stated that the pandemic situation and the measures for containing it continued to influence the trend of road accidents and mobility in 2021. Compared to the previous year, accidents and injuries increased substantially in Italy in March-June 2021, returning to levels very close to those of the pre-pandemic period in the second half of the year. As per the same source, in the year 2021, around 204.7 thousand were injured (+28.6%), and 151.9 thousand is the number of road accidents number (+28.4%). All values increased in comparison to the previous year. Thus, the increasing number of road accidents is increasing the number of trauma and hospitalizations, which is expected to increase the utilization of IO infusion kits for the rapid delivery of medications and contribute to market growth.
- Additionally, data from the Road Safety Observatory published in February 2022 stated that in January 2022, around 260 fatalities occurred on the roads of mainland France. The number of injury accidents recorded by police forces was 3,728 in January 2022, which was higher than that of 2021, which accounted for 3,508 accidents. A large number of road accidents are associated with severe injury, which requires immediate nutrients and medications, thereby boosting the demand for intraosseous infusion kits in hospitals for treating such cases.
- Therefore, owing to the aforementioned factors, the segment is expected to register a significant growth rate during the forecast period.
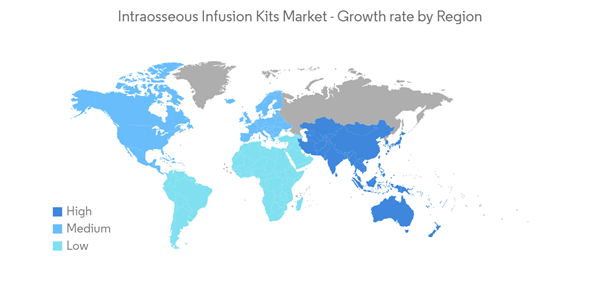
North America is Expected to Dominate the Intraosseous Infusion Kits Market Over the Forecast Period
- North America is expected to dominate the market owing to factors such as the high burden of arthritis and trauma cases such as burns, increased research and development (R&D) spending, rising awareness about the IO infusion treatment, and the high concentration of market players in the region.
- According to Statistics Canada's August 2022 update, 657.9 thousand people aged 35 to 49 years, 2 million aged 50 to 64, and 3.1 million people aged 65 and above had arthritis in 2021. Also, as per the Arthritis Foundation 2022 report, nearly 300 thousand children in the United States had juvenile arthritis. 36% of the surveyed children reported severe discomfort, significantly limiting their daily activities. These statistics indicate the high demand for IO infusion kits for the administration of medications in the distal femur or distal and proximal tibia in such diseases where another route of administration is not effective. Such factors are expected to boost market growth in the region during the forecast period.
- Furthermore, according to the National Institute of Health, Estimates of Funding for Various Research, Condition, and Disease Categories (RCDC), May 2022 update, the spending on R&D in the United States for arthritis was USD 312 million in 2021 and USD 324 million in 2022. The grant provided by the government for the research of arthritis is expected to create opportunities for market players to develop innovative devices with effective intraosseous routes of administration, which is further expected to propel market growth in the upcoming years.
- Therefore, owing to the above-mentioned factors, such as the high burden of arthritis, rising R&D spending, and high concentration of market players, the growth of the studied market is anticipated in the North American region.
Intraosseous Infusion Kits Industry Overview
The intraosseous infusion kits market is competitive in nature due to the presence of a few companies operating globally as well as regionally. The competitive landscape includes an analysis of a few international as well as local companies that hold major or significant market shares. Some market players are Aero Healthcare, BD (Becton, Dickinson and Company), BPB Medica - Biopsybell, Cook Medical, PAVmed Inc., Performance Systems, Inc. (Persys Medical), Teleflex (Pyng Medical), and Istem Medikal.Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
This product will be delivered within 2 business days.