Speak directly to the analyst to clarify any post sales queries you may have.
10% Free customizationThis report comes with 10% free customization, enabling you to add data that meets your specific business needs.
According to the United States Pharmacopeia (USP), India held the largest portion of total active API Drug Master Files (DMFs) in 2024 at 48%, with China accounting for 18%, highlighting substantial regional manufacturing capabilities.\nDespite this positive trajectory, the market encounters a major obstacle regarding supply chain stability. The concentration of API production in specific geographic areas, compounded by geopolitical dynamics, introduces vulnerabilities that can cause potential drug shortages. Furthermore, increasingly strict regulatory mandates for controlling impurities, such as those related to nitrosamines, contribute to higher compliance costs and prolonged product development cycles for global manufacturers.
Market Driver
The mounting impact of chronic and age-related illnesses directly fuels the need for Active Pharmaceutical Ingredients, as a growing number of people require long-term therapies and medications. The rising worldwide prevalence of health conditions like respiratory ailments, diabetes, and cardiovascular diseases demands a continuous and increased supply of APIs for both novel and existing drug formulations. As populations grow older and lifestyle factors encourage the onset of chronic diseases, the pharmaceutical sector is responding by boosting the output of vital medicines.The World Health Organization estimates that by May 2025, nearly 75% of the global population will be living with at least one chronic condition, creating persistent upward pressure on API manufacturing and driving capacity expansions to satisfy escalating healthcare demands.\nAnother crucial element driving the global API market is the surging demand for generic medications and biosimilars.
When patents for original drugs expire, the introduction of more budget-friendly generic and biosimilar alternatives vastly improves patient access to essential treatments. This enhanced availability, spurred by cost efficiency and supportive reimbursement frameworks, translates directly into higher manufacturing volumes for the relevant APIs. According to a GoodRx report in October 2025, the FDA had approved a minimum of 63 first-time generics by late September 2025. The competition introduced by these alternatives encourages API producers to develop more cost-effective manufacturing techniques, a dynamic that also bolsters broader industry funding, as evidenced by the 16 largest pharmaceutical firms investing a combined $159 billion in research and development during 2025.
Market Challenge
Supply chain resilience remains a formidable hurdle for the global Active Pharmaceutical Ingredient market. The localized concentration of API manufacturing in a few specific geographic areas, worsened by geopolitical tensions, establishes significant structural vulnerabilities. Relying too heavily on particular regions for vital biologically active components implies that local disturbances, including unforeseen events or trade restrictions, can rapidly escalate into extensive supply chain bottlenecks throughout the pharmaceutical industry.\nThese inherent weaknesses actively obstruct market progress by triggering severe medication shortages.Such deficits interfere with the production of final pharmaceutical goods, restricting patient access to necessary treatments and amplifying operational difficulties for pharmaceutical manufacturers across the globe. Data from the American Hospital Association indicates that 270 medications were officially listed as in short supply during the first quarter of 2025. This directly impedes the steady distribution and availability of pharmaceutical items, thereby restricting the broader growth and dependability of the Active Pharmaceutical Ingredient sector.
Market Trends
The Global Active Pharmaceutical Ingredient market is being profoundly transformed by the integration of biotechnology and personalized healthcare. This trend is propelling the demand for complex, large-molecule APIs, which require highly specialized manufacturing and developmental methodologies. In contrast to conventional small molecules, personalized therapies and biologics depend on cell-based platforms, advanced purification techniques, and rigorous handling to preserve stability. Shifting toward tailored care results in smaller, higher-value production batches, prompting manufacturers to invest in flexible, state-of-the-art facilities.The Personalized Medicine Coalition noted in May 2025 that the FDA authorized 18 new personalized treatments alongside six novel cell and gene-based therapies in 2024, underscoring the expansion of this therapeutic pipeline.\nSimultaneously, a rising necessity for complex and High-Potency APIs (HPAPIs), especially within the field of oncology, represents another crucial market trend. Because HPAPIs are highly effective at minimal doses, they mandate strict containment measures and specialized equipment to safeguard operators and prevent cross-contamination.
Fulfilling these requirements necessitates substantial capital investments in dedicated production suites, advanced analytical tools, and rigorous quality control processes. Producers are compelled to adopt heightened safety protocols and customized facility layouts; for instance, in June 2025, Piramal Pharma Solutions revealed a $90 million investment to expand its antibody-drug conjugate production, which includes HPAPI manufacturing, across two United States facilities. This illustrates the industry's proactive response to these vital active pharmaceutical ingredient manufacturing demands.
Key Market Players
- Teva Pharmaceutical Industries Ltd.
- Bristol-Myers Squibb Company
- Boehringer Ingelheim GmbH
- Merck & Co., Inc.
- GlaxoSmithKline Plc
- Novartis International AG
- F. Hoffmann-La Roche AG
- Sanofi S.A.
- Eli Lilly and Company
- AbbVie Inc.
Report Scope
In this report, the Global Active Pharmaceutical Ingredient Market has been segmented into the following categories, in addition to the industry trends which have also been detailed below:Active Pharmaceutical Ingredient Market, By Type of Synthesis:
- Biotech
- Fermentation
- Plant Extracts
- Synthetic
Active Pharmaceutical Ingredient Market, By Application:
- Cardiovascular Disease
- Oncology
- Neurological Disorders
- Orthopedic Disorders
- Respiratory
- Gastrointestinal Disorders
- Urology
- Others
Active Pharmaceutical Ingredient Market, By Region:
- North America
- Europe
- Asia Pacific
- South America
- Middle East & Africa
Competitive Landscape
Company Profiles: Detailed analysis of the major companies present in the Global Active Pharmaceutical Ingredient Market.Available Customizations
With the given market data, the publisher offers customizations according to a company's specific needs. The following customization options are available for the report:Company Information
- Detailed analysis and profiling of additional market players (up to five).
This product will be delivered within 1-3 business days.
Table of Contents
Companies Mentioned
- Teva Pharmaceutical Industries Ltd.
- Bristol-Myers Squibb Company
- Boehringer Ingelheim GmbH
- Merck & Co., Inc.
- GlaxoSmithKline Plc
- Novartis International AG
- F. Hoffmann-La Roche AG
- Sanofi S.A.
- Eli Lilly and Company
- AbbVie Inc.
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 177 |
| Published | May 2026 |
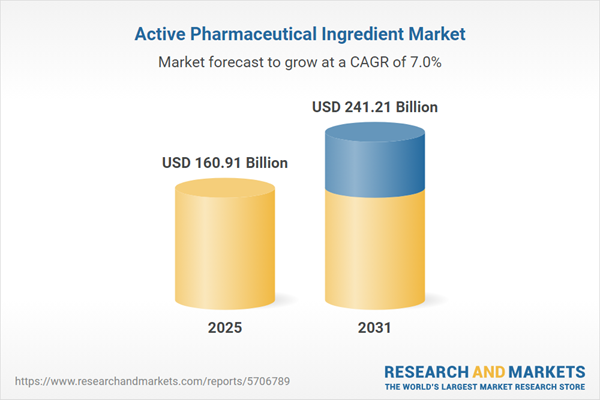
| Forecast Period | 2025 - 2031 |
| Estimated Market Value ( USD | $ 160.91 Billion |
| Forecasted Market Value ( USD | $ 241.21 Billion |
| Compound Annual Growth Rate | 6.9% |
| Regions Covered | Global |
| No. of Companies Mentioned | 10 |