Speak directly to the analyst to clarify any post sales queries you may have.
10% Free customizationThis report comes with 10% free customization, enabling you to add data that meets your specific business needs.
On the other hand, the market faces notable obstacles due to the possibility of user mistakes and issues regarding test accuracy, which can generate false results and weaken consumer trust. Consequently, individuals may be forced to seek additional medical advice or purchase multiple tests to verify their condition. Highlighting the massive economic footprint of the overarching diagnostics industry, MedTech Europe reported that the European In Vitro Diagnostics (IVD) market - which includes pregnancy test kits - achieved total revenues of €13.3 billion in 2023.
Market Driver
The Global market for pregnancy detection kits is heavily propelled by technological upgrades that improve product accuracy and overall user experience. Recent innovations have introduced highly sensitive tests that deliver clearer results and identify pregnancies much sooner. For example, data from the Hope Women's Center in April 2024 indicates that digital options like the Clearblue Early Digital Pregnancy Test can recognize human chorionic gonadotropin (hCG) levels as low as 10 mIU/ml, significantly outperforming the 25 mIU/ml standard of conventional tests to provide earlier positive readings. By minimizing user error and alleviating doubts about reliability, these advancements make home testing a highly trusted choice for early pregnancy verification.Furthermore, increasing rates of infertility and unplanned pregnancies act as a major catalyst, ensuring steady demand for discreet and easily accessible testing options. The widespread nature of these reproductive circumstances requires dependable, readily available detection tools, thereby boosting market expansion across diverse populations. Emphasizing this ongoing necessity, the World Health Organization noted in December 2025 that roughly 73 million induced abortions take place annually around the globe due to unintended pregnancies. The vast scale of the wider diagnostics industry further mirrors this high demand, evidenced by Roche Diagnostics distributing 29 billion tests to global consumers in 2023 alone.
Market Challenge
The Global market for pregnancy detection kits faces a major hurdle in the form of accuracy doubts and the likelihood of user mistakes during application. These issues can yield false negative or positive readings, severely eroding consumer faith in the efficacy of at-home diagnostic products. Encountering flawed results damages the perceived dependability and convenience of self-testing, which ultimately deters initial buyers and negatively impacts long-term brand loyalty.Consequently, a lack of trust forces users to buy additional tests or schedule clinical visits, introducing unforeseen expenses and frustrating inconveniences. Such negative experiences undermine the core benefits of privacy and immediate access that make these kits appealing in the first place. While the overarching medical technology industry boasts massive financial strength - with MedTech Europe estimating the European medtech market at roughly €170 billion in 2024 - the pregnancy kit niche remains uniquely vulnerable to accuracy-related skepticism. This skepticism acts as a direct barrier to market expansion, restricting consumer uptake and repeat sales, which ultimately prevents the sector from reaching its maximum potential within the broader diagnostics field.
Market Trends
A major trend transforming the global pregnancy detection kits market is the incorporation of smart connectivity and digital features into testing devices. Modern solutions now exceed basic visual indicators by syncing with smartphone apps to help interpret results, track fertility windows, monitor menstrual cycles, and deliver customized health data. These intelligent devices improve the overall user experience by lowering the risk of misreading outcomes and creating shareable digital logs for medical professionals. This evolution transitions the traditional pregnancy test into a comprehensive digital ecosystem for managing women's reproductive health. Highlighting the strong financial backing for such innovations, the World Economic Forum reported in January 2026 that women's health startups secured $2.6 billion in venture capital during 2024, reflecting massive investor enthusiasm for advanced diagnostic and health technologies.Additionally, the rise of subscription-based services and e-commerce distribution is heavily influencing the market landscape for pregnancy detection kits. By avoiding conventional retail stores, this model perfectly aligns with the modern consumer's desire for privacy, convenience, and direct purchasing. Digital storefronts equip buyers with comprehensive product details, customer reviews, and a broader variety of options, allowing them to make educated decisions discreetly. For consumers actively attempting to conceive, subscription models ensure a consistent delivery of tests while simultaneously creating reliable revenue streams and brand loyalty for manufacturers. Reinforcing this digital shift, Clearco noted in October 2024 that the health and wellness e-commerce market is projected to see a 20% annual increase in online sales through 2026, showcasing a robust preference for buying health supplies online.
Key Market Players
- Abbott Laboratories
- F. Hoffmann-La Roche Ltd.
- Siemens Healthineers AG
- Danaher Corporation
- Becton, Dickinson and Company (BD)
- QuidelOrtho Corporation
- bioMérieux SA
- EKF Diagnostics Holdings plc
- Cardinal Health, Inc.
- Piramal Enterprises Limited
Report Scope
In this report, the Global Pregnancy Detection Kits Market has been segmented into the following categories, in addition to the industry trends which have also been detailed below:Pregnancy Detection Kits Market, By Product Type:
- Line Indicators
- Digital Devices
- Others
Pregnancy Detection Kits Market, By Type of Test:
- Urine Test for HCG
- Blood Test for HCG
Pregnancy Detection Kits Market, By Distribution Channel:
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
Pregnancy Detection Kits Market, By Region:
- North America
- Europe
- Asia Pacific
- South America
- Middle East & Africa
Competitive Landscape
Company Profiles: Detailed analysis of the major companies present in the Global Pregnancy Detection Kits Market.Available Customizations
With the given market data, the publisher offers customizations according to a company's specific needs. The following customization options are available for the report:Company Information
- Detailed analysis and profiling of additional market players (up to five).
This product will be delivered within 1-3 business days.
Table of Contents
Companies Mentioned
- Abbott Laboratories
- F. Hoffmann-La Roche Ltd.
- Siemens Healthineers AG
- Danaher Corporation
- Becton, Dickinson and Company (BD)
- QuidelOrtho Corporation
- bioMérieux SA
- EKF Diagnostics Holdings plc
- Cardinal Health, Inc.
- Piramal Enterprises Limited
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 180 |
| Published | May 2026 |
| Forecast Period | 2025 - 2031 |
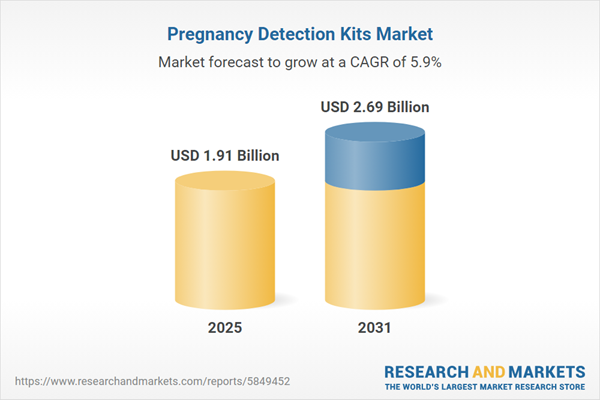
| Estimated Market Value ( USD | $ 1.91 Billion |
| Forecasted Market Value ( USD | $ 2.69 Billion |
| Compound Annual Growth Rate | 5.8% |
| Regions Covered | Global |
| No. of Companies Mentioned | 10 |