Speak directly to the analyst to clarify any post sales queries you may have.
10% Free customizationThis report comes with 10% free customization, enabling you to add data that meets your specific business needs.
Furthermore, advancements in technology, including cell culture systems, analytical tools, and manufacturing processes, are contributing to the efficiency and scalability of biologics production. CDOs are at the forefront of adopting and implementing these technologies, ensuring that they can meet the growing demand for biologics on a global scale. Additionally, the COVID-19 pandemic has highlighted the importance of a resilient and flexible biomanufacturing infrastructure, prompting increased investments in the biopharmaceutical sector and, consequently, the contract development market.
The Global Biologics Contract Development Market is characterized by a competitive landscape with numerous CDOs vying for market share. Companies in this space differentiate themselves by offering comprehensive solutions, state-of-the-art facilities, and a strong regulatory track record. Geographically, North America and Europe have traditionally been significant markets for biologics development, owing to the concentration of pharmaceutical and biotechnology companies. However, emerging markets in Asia, particularly China and India, are gaining traction due to their growing capabilities, cost advantages, and strategic investments in biopharmaceutical infrastructure.
Key Market Drivers
Growing Demand for Biopharmaceuticals
The Global Biologics Contract Development Market is experiencing a substantial boost due to the growing demand for biopharmaceuticals. Biopharmaceuticals, which encompass a diverse range of therapeutic proteins, monoclonal antibodies, vaccines, and gene therapies, have emerged as pivotal components in the treatment of various diseases, including cancer, autoimmune disorders, and infectious diseases. The escalating demand for these advanced therapies is fueled by their targeted and personalized approach, offering enhanced efficacy with fewer side effects compared to traditional pharmaceuticals.As the healthcare landscape increasingly shifts towards precision medicine, biopharmaceuticals are at the forefront of innovation, addressing unmet medical needs and providing new treatment modalities. This surge in demand has placed significant pressure on biopharmaceutical companies to expedite the development and commercialization of these complex therapies. In response, companies are turning to specialized entities, such as contract development organizations (CDOs), to streamline and accelerate the drug development process.
CDOs play a crucial role in meeting the heightened demand for biopharmaceuticals by offering specialized expertise and infrastructure for various stages of drug development, from early-phase research to commercial manufacturing. The complexity involved in the development of biologics, including intricate manufacturing processes and the need for stringent quality control, makes CDOs indispensable partners for biopharmaceutical companies seeking to navigate this intricate landscape efficiently.
Furthermore, the growing prevalence of chronic diseases, coupled with an aging global population, has contributed to an increased need for advanced therapeutic solutions. Biopharmaceuticals, with their ability to target specific disease pathways and mechanisms, are well-positioned to address these healthcare challenges. This heightened demand has led to a surge in outsourcing strategies, with biopharmaceutical companies leveraging the expertise of CDOs to harness their specialized capabilities and accelerate the development of biologics.
Advancements in Biotechnology
Advancements in biotechnology are serving as a powerful catalyst for the remarkable growth of the Global Biologics Contract Development Market. This market expansion is intricately linked to the revolutionary progress made in various facets of biotechnology, ranging from cell culture systems to gene editing technologies. These breakthroughs are instrumental in enhancing the efficiency, scalability, and precision of biologics development, positioning contract development organizations (CDOs) as essential partners in the biopharmaceutical sector. The adoption of state-of-the-art technologies enables CDOs to offer cutting-edge solutions, addressing the evolving needs of biopharmaceutical companies.Cell culture systems have witnessed significant improvements, allowing for the optimized production of biologics on a larger scale. This not only meets the surging global demand for biopharmaceuticals but also contributes to the cost-effective and timely delivery of these advanced therapies. Additionally, innovations in gene editing technologies, such as CRISPR-Cas9, have revolutionized the field by facilitating precise modifications in cellular structures. This level of precision is particularly crucial in the development of gene therapies and personalized medicines, further fueling the demand for CDOs with expertise in these cutting-edge technologies.
Analytical tools have also undergone substantial advancements, enabling more robust characterization and quality control of biologics. CDOs equipped with these sophisticated analytical capabilities can ensure the integrity, purity, and consistency of biopharmaceutical products throughout the development and manufacturing processes. This, in turn, instills confidence in regulatory compliance, a critical aspect of the biologics development landscape.
Furthermore, the integration of automation and digital technologies in biomanufacturing processes is transforming the production efficiency and agility of CDOs. These advancements contribute to the flexibility required to adapt to the dynamic demands of the biopharmaceutical industry, facilitating rapid scale-up or scale-down as needed. The enhanced efficiency resulting from these technological integrations is pivotal in meeting the stringent timelines associated with biologics development.
Complexity of Biologics Development
The complexity inherent in biologics development stands as a driving force behind the flourishing Global Biologics Contract Development Market. Biologics, encompassing a diverse array of therapeutic proteins, monoclonal antibodies, vaccines, and gene therapies, pose unique challenges compared to traditional small-molecule drugs. The intricate nature of biologics necessitates specialized expertise in various critical aspects, including cell line development, process optimization, and analytical testing, making the involvement of contract development organizations (CDOs) paramount.Cell line development, a crucial early phase in biologics development, requires the identification and cultivation of cell lines capable of producing the desired therapeutic proteins. This process demands precision and a deep understanding of cellular biology, and CDOs specializing in biologics excel in navigating these complexities. They offer specialized knowledge and infrastructure to optimize cell lines for maximum productivity and desired product attributes.
Process optimization, another intricate facet, involves refining the manufacturing processes to ensure efficiency, yield, and product quality. CDOs play a pivotal role in streamlining these processes, leveraging their experience and capabilities to enhance scalability and reproducibility. The ability to navigate the intricacies of process optimization positions CDOs as indispensable partners in overcoming challenges associated with the large-scale production of biologics.
Analytical testing in biologics development requires sophisticated methodologies to assess product quality, safety, and efficacy. The complexity of biologic molecules demands advanced analytical techniques, and CDOs are equipped with state-of-the-art analytical tools to meet these stringent requirements. Their expertise in analytical testing ensures the thorough characterization of biopharmaceutical products, providing critical data for regulatory submissions and quality assurance.
Moreover, the regulatory landscape governing biologics is rigorous and continually evolving, necessitating a deep understanding of global regulatory standards. CDOs specializing in biologics contract development bring invaluable regulatory expertise, guiding biopharmaceutical companies through the intricate process of compliance and approval. This regulatory proficiency is a key factor in the successful progression of biologics from development to commercialization.
Key Market Challenges
Regulatory Uncertainty
Regulatory uncertainty poses a significant challenge to the Global Biologics Contract Development Market, influencing the dynamics of biopharmaceutical development and the role of contract development organizations (CDOs). The intricate nature of biologics, including therapeutic proteins, monoclonal antibodies, vaccines, and gene therapies, necessitates stringent regulatory oversight to ensure product safety, efficacy, and quality. However, the evolving regulatory landscape introduces uncertainties that impact timelines, costs, and strategic decision-making for both CDOs and biopharmaceutical companies. One of the primary challenges within regulatory uncertainty is the lack of harmonization across global regulatory agencies. Divergent regulations and varying expectations in different regions complicate the development and approval processes for biologics. CDOs operating on a global scale must navigate a complex web of regulatory requirements, which can lead to delays and increased compliance costs. This lack of uniformity adds an additional layer of complexity, requiring CDOs to maintain a deep understanding of regulatory frameworks across multiple jurisdictions.Furthermore, the dynamic nature of regulatory guidelines for biologics presents an ongoing challenge. Regulatory agencies continually update and revise standards to keep pace with scientific advancements and address emerging issues. This frequent evolution requires CDOs to stay agile and adapt quickly to changes in requirements, which can be resource-intensive. The uncertainty regarding upcoming regulatory shifts may impact project planning, resource allocation, and overall timelines, introducing an element of unpredictability in the biologics contract development process.
Quality Control and Analytical Challenges
Quality control and analytical challenges stand out as formidable hurdles in the Global Biologics Contract Development Market, impacting the development and manufacturing of complex biopharmaceuticals. The unique attributes of biologics, such as monoclonal antibodies, therapeutic proteins, and gene therapies, demand rigorous quality control measures to ensure product safety, efficacy, and consistency. These challenges pose complexities that must be navigated by contract development organizations (CDOs) to meet stringent regulatory standards and ensure the success of biologics development projects.One of the primary challenges lies in the intricacies of characterizing and analyzing biologic molecules. Unlike small-molecule drugs, biologics exhibit structural diversity and complexity, requiring advanced analytical techniques for accurate assessment. Ensuring the reproducibility and consistency of biologic products throughout development and manufacturing becomes a daunting task, demanding a continuous refinement of analytical methodologies to meet evolving regulatory expectations.
Biologics, being produced from living systems, are subject to inherent variability. This inherent variability can introduce challenges in maintaining consistent product quality. Analytical methods must be capable of detecting and quantifying subtle variations in product attributes, such as glycosylation patterns and protein folding, which can impact the safety and efficacy of the final product. The complexity of biologics necessitates the development and validation of sophisticated analytical tools that can provide comprehensive insights into product quality and attributes.
Key Market Trends
Increasing Outsourcing of Biologics Development
The Global Biologics Contract Development Market is experiencing a significant boost due to the increasing outsourcing of biologics development by pharmaceutical and biotechnology companies. As the demand for complex biologics, including therapeutic proteins, monoclonal antibodies, and gene therapies, continues to rise, companies are recognizing the advantages of leveraging the expertise and specialized services offered by contract development organizations (CDOs). Outsourcing biologics development allows pharmaceutical companies to streamline their operations, reduce time-to-market, and allocate resources more efficiently. CDOs play a pivotal role in this trend by offering a comprehensive range of services, covering various stages of biologics development, from early research and development to commercial manufacturing.The complexities inherent in biologics development, such as intricate manufacturing processes and the need for specialized analytical testing, make outsourcing a strategic choice for many companies. CDOs bring a wealth of experience and infrastructure dedicated to biologics, ensuring that pharmaceutical companies can tap into specialized knowledge without having to build and maintain these capabilities in-house. This strategic shift toward outsourcing enables pharmaceutical companies to focus on their core competencies while benefiting from the efficiencies and expertise offered by CDOs.
Moreover, outsourcing provides a flexible and scalable approach to biologics development. As the biopharmaceutical industry experiences dynamic changes and fluctuations in demand, companies can leverage the agility of CDOs to adapt to these shifts. This flexibility is particularly crucial in the development of biologics, where factors like cell line development, process optimization, and analytical testing require specialized attention and can benefit from the dedicated resources of CDOs.
Focus on Personalized Medicine
The Global Biologics Contract Development Market is experiencing a notable surge propelled by the industry's intensified focus on personalized medicine. As the understanding of individualized patient responses to treatments deepens, biopharmaceutical companies are increasingly tailoring their therapeutic approaches to meet the unique characteristics of patients. This paradigm shift towards personalized medicine is particularly pronounced in the development of biologics, including therapeutic proteins, monoclonal antibodies, vaccines, and gene therapies. Contract development organizations (CDOs) are playing a pivotal role in supporting this trend by providing specialized services that cater to the intricacies of personalized medicine initiatives.Biologics, with their ability to target specific disease pathways and mechanisms, are well-suited for personalized treatment strategies. The customization of biologic therapies allows for a more precise and targeted approach, minimizing adverse effects and improving overall treatment outcomes. CDOs are responding to this demand by offering services that align with the requirements of personalized medicine, such as the development of personalized cell therapies and gene therapies that cater to individual patient profiles.
The trend towards personalized medicine is reshaping traditional drug development processes, necessitating a more nuanced understanding of patient populations and their genetic or molecular characteristics. CDOs are at the forefront of adopting innovative technologies and methodologies that enable the customization of biologic therapies based on individual patient needs. This includes advancements in cell and gene therapy development, where CDOs are contributing expertise in optimizing these personalized treatment modalities.
Additionally, the emphasis on personalized medicine is influencing clinical trial designs, with a growing number of trials incorporating biomarker-driven approaches to identify patient subpopulations that respond more favorably to specific biologics. CDOs are crucial partners in this process, contributing to the design and execution of clinical trials that align with personalized medicine principles. The increasing collaboration between biopharmaceutical companies and CDOs in the development of personalized biologics reflects a strategic alignment with the future direction of healthcare. The potential benefits of personalized medicine, including improved patient outcomes and reduced healthcare costs, are driving this transformative trend.
Segmental Insights
Source Insights
Based on the Source, Mammalian emerged as the dominant segment in the global market for Global Biologics Contract Development in 2023. Mammalian expression systems offer several advantages for the production of complex biologics, such as monoclonal antibodies and therapeutic proteins. Mammalian cells provide the necessary cellular machinery for the correct folding, assembly, and post-translational modifications of complex proteins. This ensures that the produced biologics closely resemble those found in the human body, reducing the risk of immunogenicity and enhancing the efficacy and safety of the final therapeutic product. The high demand for mammalian cell expression systems is driven by the increasing complexity of biologics in development, especially those requiring precise glycosylation patterns and other post-translational modifications. Additionally, mammalian cells are preferred for the production of biologics intended for use in humans due to their ability to generate proteins with appropriate structures and functions.Indication Insights
Based on the Indication, Oncology emerged as the dominant segment in the global market for Global Biologics Contract Development Market in 2023. Many biologics, including monoclonal antibodies, immunotherapies, and targeted therapies, are developed for various types of cancer. The complex nature of cancer and the need for highly targeted and personalized treatments make biologics particularly well-suited for oncology. Immunotherapies, in particular, have gained prominence in oncology. Monoclonal antibodies that target specific cancer cells or enhance the body's immune response against cancer have shown significant clinical success. The demand for innovative biologics to treat different types of cancers continues to grow, contributing to a high demand for biologics contract development services in the oncology space.Regional Insights
North America emerged as the dominant player in the Global Biologics Contract Development Market in 2023, holding the largest market share. North America, particularly the United States, has traditionally been a major hub for biopharmaceutical research, development, and manufacturing. The region hosts numerous biotechnology and pharmaceutical companies, research institutions, and contract development organizations (CDOs) specializing in biologics. The United States, in particular, is home to a significant concentration of biopharmaceutical companies and CDOs. This concentration fosters a collaborative ecosystem that drives innovation and expertise in biologics development. North America leads in biopharmaceutical research and development, with a robust pipeline of biologics in various stages of development. This innovation contributes to the demand for contract development services.Report Scope:
In this report, the Global Biologics Contract Development Market has been segmented into the following categories, in addition to the industry trends which have also been detailed below:Global Biologics Contract Development Market, By Source:
- Mammalian
- Microbial
- Others
Global Biologics Contract Development Market, By Service:
- Cell Line Development
- Process Development
- Others
Global Biologics Contract Development Market, By Indication:
- Oncology
- Immunological Disorders
- Cardiovascular Disorders
- Hematological Disorders
- Others
Global Biologics Contract Development Market, By Region:
- North America
- United States
- Canada
- Mexico
- Europe
- France
- United Kingdom
- Italy
- Germany
- Spain
- Asia-Pacific
- China
- India
- Japan
- Australia
- South Korea
- South America
- Brazil
- Argentina
- Colombia
- Middle East & Africa
- South Africa
- Saudi Arabia
- UAE
- Egypt
Competitive Landscape
Company Profiles: Detailed analysis of the major companies present in the Global Biologics Contract Development Market.Available Customizations:
Global Biologics Contract Development Market report with the given market data, the publisher offers customizations according to a company's specific needs.This product will be delivered within 1-3 business days.
Table of Contents
Companies Mentioned
- WuXi Biologics
- Abzena Ltd
- Fujifilm Diosynth Biotechnologies
- KBI Biopharma
- AGC Biologics
- Thermo Fisher Scientific Inc.
- Curia Global, Inc.
- Genscript Biotech
- Bionova Scientific, Inc.
- BioXcellence (Boehringer Ingelheim Biopharmaceuticals GmbH)
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 180 |
| Published | February 2024 |
| Forecast Period | 2023 - 2029 |
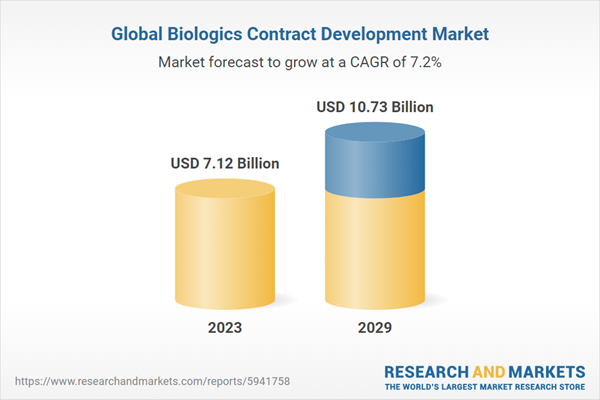
| Estimated Market Value ( USD | $ 7.12 Billion |
| Forecasted Market Value ( USD | $ 10.73 Billion |
| Compound Annual Growth Rate | 7.2% |
| Regions Covered | Global |
| No. of Companies Mentioned | 10 |