The market is experiencing steady expansion driven by the rising number of global clinical trials, continuous advancements in laboratory testing technologies, and increased investment in research and development activities across the life sciences sector. Growing outsourcing of laboratory services by hospitals and clinics is also contributing significantly to market growth. Rising disease prevalence is leading to higher test volumes, while improved logistics networks are enabling faster and more efficient sample transportation. The expansion of genomic and molecular testing is further strengthening demand for advanced central laboratory capabilities. In addition, the increasing adoption of decentralized clinical trial models and the growing participation of emerging markets in global trials are reinforcing the need for standardized, high-quality laboratory infrastructure. Technological improvements such as automation systems, high-throughput analyzers, digital pathology tools, and advanced molecular testing platforms are significantly enhancing operational efficiency, accuracy, and reproducibility. These advancements are ensuring consistent data quality across multi-site clinical studies, supporting more reliable drug development outcomes, and reinforcing the strategic importance of central laboratories in global clinical research.
The biomarker services segment accounted for 31.3% share in 2025, owing to the increasing outsourcing of diagnostic and testing services by healthcare institutions. Biomarker-based testing plays a critical role in drug development and precision medicine by supporting patient classification, therapeutic target validation, safety monitoring, and treatment response evaluation across clinical trials. The rising adoption of molecular, genetic, and protein-based biomarker testing is increasing the demand for standardized, high-precision laboratory services.
The oncology segment generated USD 1.3 billion in 2025, making it the leading application area within the central lab market. Oncology research relies heavily on complex diagnostic testing, including genomic analysis and tissue-based evaluations, which require centralized laboratory support to ensure consistency and accuracy across trial sites. The growing development of targeted therapies, immunotherapies, and cell-based treatment approaches is further increasing reliance on advanced laboratory services. Centralized oncology testing enhances data reliability, improves patient selection processes, and supports adaptive clinical trial designs, making it essential for modern cancer research programs.
U.S. Central Lab Market was valued at USD 1.4 billion in 2025. Market expansion in the country is supported by rising diagnostic demand driven by increasing disease prevalence and the growing adoption of automation to improve laboratory efficiency and operational performance. Strong emphasis on personalized medicine is also boosting demand for advanced diagnostic and biomarker testing services. Continued investment from both government and private sectors in biomedical research and clinical trials is further strengthening market growth. In addition, the rising burden of chronic conditions such as cancer and diabetes is increasing the need for comprehensive testing services, supporting sustained demand for central laboratory infrastructure across the United States.
Key companies operating in the Global Central Lab Market include Eurofins Scientific, ICON plc, Labcorp, IQVIA Laboratories, Quest Diagnostics, Thermo Fisher Scientific, Parexel International Corporation, Medpace, Inc., Cerba Research, ACM Global Laboratories, BioAgilytix Labs, Frontage Laboratories, LabConnect, Lambda Therapeutic Research, Medicover Integrated Clinical Services (MICS), and Versiti. Companies in the Central Lab Market are focusing on strengthening their market presence through continuous investment in advanced laboratory automation, digital transformation, and high-throughput testing capabilities. Many players are expanding their global laboratory networks to support increasing clinical trial activity across multiple regions. Strategic partnerships with pharmaceutical and biotechnology companies are enabling improved integration into drug development pipelines. Firms are also enhancing their service portfolios by incorporating advanced biomarker testing, genomic analysis, and specialized diagnostic solutions. In addition, companies are prioritizing data management systems and digital platforms to improve accuracy, traceability, and real-time reporting.
Comprehensive Market Analysis and Forecast
- Industry trends, key growth drivers, challenges, future opportunities, and regulatory landscape
- Competitive landscape with Porter’s Five Forces and PESTEL analysis
- Market size, segmentation, and regional forecasts
- In-depth company profiles, business strategies, financial insights, and SWOT analysis
This product will be delivered within 2-4 business days.
Table of Contents
Companies Mentioned
The companies profiled in this Central Lab market report include:- ACM Global Laboratories
- BioAgilytix Labs
- Cerba Research
- Eurofins Scientific
- Frontage Laboratories
- ICON plc
- IQVIA Laboratories
- LabConnect
- Labcorp
- Lambda Therapeutic Research
- Medicover Integrated Clinical Services (MICS)
- Medpace, Inc.
- Parexel International Corporation
- Quest Diagnostics
- Thermo Fisher Scientific
- Versiti
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 135 |
| Published | April 2026 |
| Forecast Period | 2025 - 2035 |
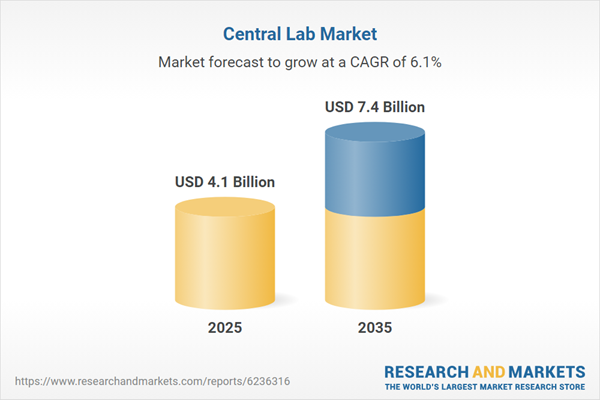
| Estimated Market Value ( USD | $ 4.1 Billion |
| Forecasted Market Value ( USD | $ 7.4 Billion |
| Compound Annual Growth Rate | 6.1% |
| Regions Covered | Global |
| No. of Companies Mentioned | 17 |