Speak directly to the analyst to clarify any post sales queries you may have.
10% Free customizationThis report comes with 10% free customization, enabling you to add data that meets your specific business needs.
Growth in this sector is largely fueled by rising global requirements for parenteral therapeutics, specifically vaccines and biologics, alongside stringent regulatory enforcement of Good Manufacturing Practices that demand superior sterility assurance. Additionally, the industry is motivated by the necessity to reduce human involvement via automated solutions. In 2024, the Association for Packaging and Processing Technologies reported that 83% of pharmaceutical manufacturers rank equipment reliability and repeatability as their primary concerns when selecting new machinery, highlighting the urgent need for high-performance systems that satisfy these exacting standards.
Despite this positive growth outlook, the market encounters significant hurdles related to the extensive capital expenditures needed for advanced aseptic technologies. Installing and validating intricate containment systems, including isolators and restricted access barrier systems, requires substantial upfront investment and specialized technical knowledge, which can discourage smaller manufacturers with tighter budgets from modernizing their infrastructure. As a result, these financial and operational obstacles may hinder the widespread adoption of next-generation processing solutions, potentially retarding overall market development in emerging regions.
Market Drivers
The escalating demand for sterile injectables and parenteral therapeutics is fundamentally transforming the sector, forcing manufacturers to rapidly expand their aseptic processing capacities. This momentum is especially visible in the competitive rush to secure fill-finish capabilities for high-demand biologics, such as GLP-1 receptor agonists, which require the deployment of high-speed, contamination-free filling lines. Prominent pharmaceutical companies are actively acquiring contract manufacturing resources to ensure supply chain stability and sterility assurance for these delicate products. For instance, in a strategic effort to bolster sterile manufacturing capacity for its diabetes and weight-loss portfolios, Novo Holdings announced in February 2024 that it agreed to acquire Catalent in an all-cash transaction with an enterprise value of $16.5 billion.At the same time, the incorporation of advanced robotics and Industry 4.0 automation serves as a vital driver for equipment adoption, spurred by the necessity to eradicate human error within sterile settings. Modern aseptic facilities are evolving toward fully automated material handling systems and gloveless robotic isolators that ensure adherence to rigorous regulatory standards while optimizing throughput.
This transition is underpinned by widespread industry commitment to digital transformation aimed at improving operational efficiency and batch uniformity. According to Rockwell Automation’s "State of Smart Manufacturing: Life Sciences Edition" report from March 2024, 96% of life sciences manufacturers are currently utilizing or assessing smart manufacturing technologies. This wave of technological upgrades aligns with massive capital projects, such as Eli Lilly's 2024 announcement of an additional $5.3 billion investment to expand its Indiana manufacturing site, underscoring the immense scale of new facilities demanding advanced aseptic machinery.
Market Challenges
The Global Aseptic Pharma Processing Equipment Market faces significant constraints due to the massive capital expenditures necessary to procure and validate advanced containment technologies. Deploying systems like restricted access barrier systems and isolators demands a heavy upfront investment that encompasses not only the purchase of machinery but also expensive facility retrofitting and specialized utility installations. This financial strain is further exacerbated during the rigorous validation phase, where manufacturers must bear extra costs to prove compliance with stringent sterility standards before production operations can begin.These economic pressures establish a formidable barrier to entry that impedes the widespread adoption of automated processing solutions, especially among companies in emerging economies and smaller contract manufacturing organizations. The prohibitive total cost of ownership effectively prevents budget-constrained entities from updating their facilities, thereby slowing the industry-wide transition toward automated sterility assurance. According to the International Society for Pharmaceutical Engineering, nearly 50% of industry professionals surveyed in 2024 pinpointed regulatory complexities and economic factors as the main obstacles to adopting innovative manufacturing technologies. This evidence highlights how financial limitations directly restrict market growth by deterring investment in essential modernization.
Market Trends
The transition toward modular and flexible manufacturing facilities is altering investment strategies as pharmaceutical firms value agility over dedicated single-product lines to manage diverse pipelines. In contrast to traditional fixed stainless-steel lines built for high-volume mono-production, contemporary facilities are incorporating adaptable processing modules that facilitate rapid product changeovers and the concurrent manufacturing of various biologic modalities. This operational flexibility is crucial for manufacturers aiming to balance production between blockbuster drugs and lower-volume therapeutics without the need to build separate plants for each. A prime example of this strategic shift occurred in May 2024, when Sanofi committed over €1 billion to establish new bioproduction capacity in France, specifically designing its Vitry-sur-Seine site to flexibly manufacture treatments for diverse indications, including multiple sclerosis, asthma, and COPD.Concurrently, the development of small-batch processing equipment for personalized medicine is gathering momentum, fueled by the commercialization of autologous cell and gene therapies that demand highly contained, low-volume manufacturing settings. Since these patient-specific treatments cannot be processed on high-speed filling lines, the market is seeing an increase in specialized, closed-system equipment engineered to manage micro-batches with zero risk of cross-contamination. This trend compels equipment developers to pivot from speed-focused designs to systems that emphasize containment integrity and precision for high-value biological materials. This sectoral focus was clearly demonstrated in February 2024, when AstraZeneca announced a $300 million investment in a specialized facility in Rockville, Maryland, explicitly dedicated to launching its cell therapy platforms and producing T-cell therapies for commercial supply and clinical trials.
Key Players Profiled in the Aseptic Pharma Processing Equipment Market
- Robert Bosch GmbH
- DuPont plc
- SPX Flow, Inc.
- Industria Macchine Automatiche S.P.A.
- Becton, Dickinson, and Company
- AMCOR PLC
- GEA Group
- Greatview Aseptic Packaging Co., Ltd.
- John Bean Technologies (JBT) Corporation
- Automated Systems of Tacoma, LLC
Report Scope
In this report, the Global Aseptic Pharma Processing Equipment Market has been segmented into the following categories:Aseptic Pharma Processing Equipment Market, by Component:
- Processing Equipment (Drying Equipment
- Extruders
- Mills
- Granulation Equipment
- Tablet Compression
- Feeders
- Fill/Finish Equipment
- Mixing Equipment
- Pneumatic Conveying Equipment)
- Packaging Equipment (Inspection Machines
- Labelling Machines
- Aseptic Packaging Machines)
Aseptic Pharma Processing Equipment Market, by Application:
- Ophthalmic Suspensions
- Sterile Injectables
- Reconstituted Lyophilized Powders for Injection
- Aqueous-based Aerosols for Inhalation
- Others
Aseptic Pharma Processing Equipment Market, by Region:
- North America
- Europe
- Asia-Pacific
- South America
- Middle East & Africa
Competitive Landscape
Company Profiles: Detailed analysis of the major companies present in the Global Aseptic Pharma Processing Equipment Market.Available Customization
The analyst offers customization according to your specific needs. The following customization options are available for the report:- Detailed analysis and profiling of additional market players (up to five).
This product will be delivered within 1-3 business days.
Table of Contents
Companies Mentioned
The key players profiled in this Aseptic Pharma Processing Equipment market report include:- Robert Bosch GmbH
- DuPont PLC.
- SPX Flow, Inc.
- Industria Macchine Automatiche S.P.A.
- Becton, Dickinson, and Company
- AMCOR PLC
- GEA Group
- Greatview Aseptic Packaging Co., Ltd.
- John Bean Technologies (JBT) Corporation
- Automated Systems of Tacoma, LLC
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 180 |
| Published | January 2026 |
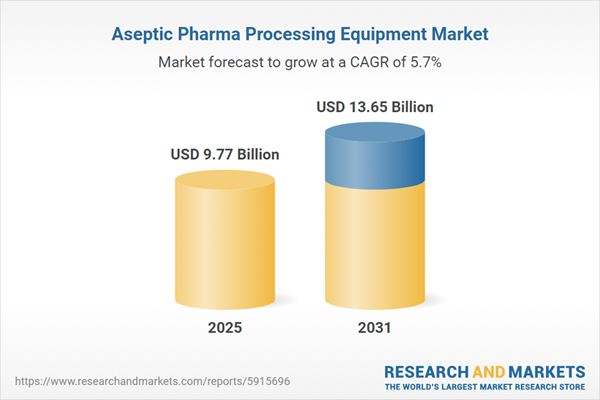
| Forecast Period | 2025 - 2031 |
| Estimated Market Value ( USD | $ 9.77 Billion |
| Forecasted Market Value ( USD | $ 13.65 Billion |
| Compound Annual Growth Rate | 5.7% |
| Regions Covered | Global |
| No. of Companies Mentioned | 11 |